| |
|
|
INTRODUCTION Chronic heart failure is a progressive and
complex syndrome representing the end stage of various
cardiovascular diseases and is one of the leading causes of
morbidity and mortality worldwide. Acute heart failure is the most
severe clinical form, characterized by cardiogenic pulmonary edema
and cardiogenic shock, with the highest mortality, requiring urgent
hospital treatment, although it is fortunately much less common than
chronic heart failure. The prevalence in the general population is
estimated at 1–2%, while in individuals older than 75 years it
reaches up to 10% [1,2]. Globally, more than 64 million people live
with this condition, and a further increase is expected due to
population aging [3].
Decompensated chronic heart failure is characterized by congestion
in the pulmonary and/or systemic circulation and fluid accumulation
in the body. The most severe form of this process is anasarca, a
diffuse generalized edema that may include ascites, pleural
effusions, and pericardial effusion [4]. In addition to heart
failure, anasarca also occurs in other conditions (nephrotic
syndrome, liver cirrhosis, severe hypoalbuminemia), but in the
context of heart failure it indicates a terminal stage, exhaustion
of compensatory mechanisms, and poor prognosis [5].
The diagnosis and management of patients with anasarca are
challenging, as a combination of cardiac, renal, and hepatic
dysfunction is often present. Treatment is based on aggressive and
individualized intravenous diuretic therapy, correction of
electrolyte imbalances, optimization of hemodynamics, and a
multidisciplinary approach involving consultants [6].
This case report is particularly significant as it demonstrates that
a severe form of heart failure, seemingly refractory terminal-stage
decompensated chronic heart failure, can be successfully treated
with intensive therapy dominated by high-dose intravenous furosemide,
in addition to the standard pillars of heart failure management in
patients with anasarca.
The contemporary approach to chronic heart failure treatment is
based on the so-called “four pillars of therapy” (ARNI/ACE
inhibitors, beta-blockers, mineralocorticoid receptor antagonists,
and SGLT2 inhibitors), which significantly reduce mortality and
hospitalizations [6–9]. Equally important in the setting of anasarca
is the “fifth pillar” — diuretic therapy with intravenous loop
diuretics.
CASE REPORT
Basic patient data: Male patient, K.A., 88 years old;
anthropometric parameters: body weight 78 kg, height 167 cm, body
mass index (BMI) 29.4 kg/m², body surface area (BSA) 1.9 m², waist
circumference 92 cm, and oxygen saturation (SpO₂) 96%.
The patient was admitted on June 6, 2025, in a state of severe
decompensated chronic heart failure with marked congestion and
anasarca, representing a clinical indicator of advanced disease [5].
Medical history: The patient presented with progressive dyspnea,
swelling of the lower legs and forearms, marked weakness, fatigue,
and shortness of breath on minimal exertion and at rest following
exertion. Symptoms had been present for the past 14 days, with rapid
progression. Increased fatigue had been noted over the previous two
weeks (he was unable to climb to the first floor), accompanied by
rapid and irregular heart rhythm and significant swelling of the
lower legs, followed by the forearms.
He was examined by an internist in the hospital one week earlier,
when low-dose therapy was initiated: furosemide 40 mg once daily
orally, spironolactone 25 mg once daily, and rivaroxaban 15 mg once
daily. The patient denied chest pain. Blood pressure at home was
generally low. He reported a history of treated hypertension over
the past four years, without prior use of cardiac medications.
Previous long-term therapy: bisoprolol 5 mg (1 + 0 + ½), rivaroxaban
15 mg once daily, ramipril/hydrochlorothiazide 5/25 mg once daily in
the morning, ramipril 5 mg once daily in the evening, allopurinol
100 mg once daily, furosemide 40 mg once daily, spironolactone 25 mg
once daily, and iron supplementation 30 mg once daily.
Physical examination on admission:
General condition: Pale and dyspneic, acyanotic, afebrile.
Vital parameters: blood pressure 110/60 mmHg, heart rate
approximately 82/min, irregular rhythm. Oxygen saturation (SpO₂)
95%. Skin and mucous membranes pale.
Lungs (auscultation): Breath sounds diminished, bilaterally
absent at the bases; percussion note dull at the lung bases on both
sides.
Heart: Displaced apical impulse on palpation. Heart rate
82/min, heart sounds attenuated, rhythm irregular consistent with
atrial fibrillation. A holosystolic regurgitant murmur grade 2–3/6
was heard over the apex, without radiation.
Abdomen: The liver was palpable 4 cm below the right costal
margin in the midclavicular line, with soft consistency; the spleen
was not palpable. No signs of ascites.
Extremities: Massive, pitting, cold edema of the lower legs
(right side: subpatellar circumference 41 cm, mid-tibial 36 cm,
supramalleolar 25 cm; left side respectively 35 cm, 34 cm, 27 cm);
mild edema of the hands and forearms.
Laboratory findings:
- NT-proBNP: 1314 pg/mL (<526 for patient’s age)
- D-dimer: 0.46 μg/mL (<0.4)
- Urea: 17 mmol/L
- Creatinine: 133 μmol/L
- GFR: 44 mL/min/1.73 m²
- Liver enzymes: AST 96 U/L, ALT 165 U/L
- Hemoglobin: 68 g/L, RBC 3.65 × 10¹²/L, MCV 67.7 fL
Laboratory findings indicated chronic heart failure with
cardiorenal syndrome and severe anemia, which are common
comorbidities and further worsen prognosis [10–12].
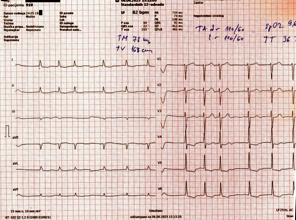
Electrocardiogram (ECG):
Atrial fibrillation with absolute ventricular arrhythmia, heart rate
82/min, intermediate electrical axis, normal QRS duration,
occasional ventricular extrasystoles (PVCs), ST depression up to 2
mm with negative T waves in leads V4–V6, and ST depression up to 0.5
mm with negative T waves in leads I, II, and aVL (Figure 1).
Figure 1 – Electrocardiogram (ECG) on admission
ECHOCARDIOGRAPHY:
The findings are dominated by left ventricular dilation, with
normal left ventricular wall thickness, no myocardial hypertrophy,
and reduced global systolic function: the left ventricular ejection
fraction (EF) was 39% by M-mode (Teichholz method) (Figure 2), and
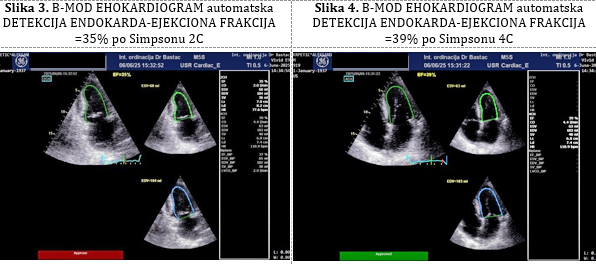
35% and 39% by Simpson’s method, with a biplane EF of 37% (Figures
3, 4).
Figure 2 – M-mode echocardiogram: ejection
fraction (EF) = 39% according to Teichholz
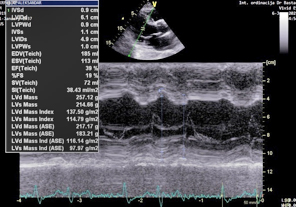
Figure 3 – B-mode echocardiogram: automatic
endocardial border detection – ejection fraction (EF) = 35% by
Simpson’s method (2-chamber view) Figure 4. – B-mode echocardiogram:
automatic endocardial border detection – ejection fraction (EF) =
39% by Simpson’s method (4-chamber view)
A small aneurysm of the basal segment was observed on the
inferior wall, with a suspected organized thrombus. Anteroapical and
anteroseptoapical dyskinesia were present. The most representative
parameter of diastolic function, the E/e′ ratio, was markedly
elevated at 15.0 (normal <8.0; E/e′ represents the ratio of early
transmitral inflow velocity (E) to the average mitral annular
velocity on tissue Doppler imaging (e′).
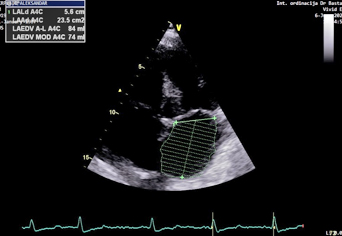
The left atrium was dilated, with a left atrial volume index (LAVI)
of 45 mL/m² (normal <34 mL/m²) (Figure 5). The maximum velocity (Vmax)
of tricuspid regurgitation was measured at 3.3 m/s (tricuspid
gradient 44 mmHg), and the right ventricular systolic pressure was
64 mmHg.
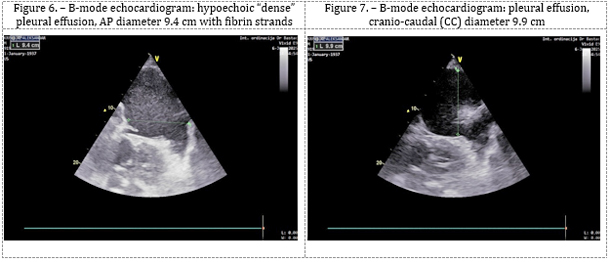
Lung ultrasound: Hydrothorax was present, with an anteroposterior
(AP), basal pleural, hypoechoic “dense” effusion: on the left side
measuring 9.9 × 9.4 cm (Figures 6, 7), with an AP diameter of 6.5 ×
4.6 cm at the level of the scapular angle; on the right side, 3 cm
below the scapular angle (AP), measuring 7.0 × 7.0 cm, and laterally
7.0 × 10.0 cm.
Figure 5 – Left atrial volume index (LAVI)
Working diagnoses:
Congestive heart failure (ICD-10: I50), with echocardiographically
reduced left ventricular ejection fraction (HFrEF ≈ 35%),
accompanied by the following cardiac conditions: permanent atrial
fibrillation (fibrillatio atriorum permanens), post-infarction
myocardial scar of the inferior wall (cicatrix myocardii post
infarctum parietis inferioris), functional left ventricular aneurysm
of the inferior wall (aneurysma functionalis ventriculi sinistri
cordis parietis inferioris), bilateral hydrothorax, mitral valve
insufficiency and aortic semilunar valve insufficiency, and
pulmonary arterial hypertension.
Comorbidities: Chronic kidney disease stage 3b (morbus renalis
gradus 3b) and severe chronic microcytic iron-deficiency anemia (anemia
microcytica sideropenica chronica, gradus gravis).
COURSE OF DISEASE (DECURSUS MORBI)
The patient refused the proposed hospitalization at the
Department of Internal Medicine, ZC Zaječar, despite being informed
about the life-threatening condition requiring intensive care
management. He was followed in a day-hospital setting at our
outpatient facility with continuous ECG monitoring, blood pressure
and urine output measurements, oxygen saturation monitoring, and
other vital parameters. Due to severe anemia, blood transfusion of
packed red blood cells was indicated; however, the patient did not
present to the Blood Transfusion Service.
The patient was immediately started on intensified parenteral
diuretic therapy: on the day of examination, furosemide ampoules 20
mg, total No VIII (160 mg), administered in two intravenous boluses,
in accordance with recommendations for the treatment of acute
decompensation [6,13,14]. Early and aggressive diuretic therapy
resulted in significant reduction of volume overload, which
represents a key therapeutic goal [13–15]. A prompt and excellent
diuresis was achieved.
On the following day, intravenous furosemide was continued at 20 mg
ampoules No IV (80 mg). Previous outpatient therapy was adjusted:
bisoprolol 5 mg tablets ½ tablet twice daily; rivaroxaban 15 mg once
daily; spironolactone 25 mg once daily; and iron supplementation.
Ramipril/hydrochlorothiazide 5/25 mg once daily in the morning,
ramipril 5 mg once daily in the evening, and allopurinol 100 mg once
daily were discontinued.
Guideline-directed medical therapy was introduced with ARNI:
sacubitril/valsartan 26/24 mg, ½ tablet twice daily, and
dapagliflozin 10 mg once daily, in accordance with current
recommendations [7,8,16–18]. For improved correction of anemia, the
iron therapy was intensified to a maximum dose of iron preparation
(300 mg/day) instead of the previous supplementation regimen.
Correction of anemia was initiated due to its negative impact on
functional status and clinical outcome, as it further aggravates
tissue hypoxia [12,19]. Non-pharmacological measures included strict
fluid and salt restriction and prohibition of physical activity.
Under this therapeutic approach, a progressive increase in diuresis
and significant reduction of edema were observed..
At the first control visit on the third day of treatment (June 7,
2025), an excellent response in fluid removal was observed: the
patient had a weight reduction of 7 kg, with complete resolution of
dyspnea on minimal exertion and a significant reduction of lower
limb edema (circumference measurements: right leg 40 cm, 35 cm, 24
cm; left leg 36 cm, 34 cm, 24 cm). Hemoglobin increased to 72 g/L.
At this point, from day 4, oral therapy was introduced with
furosemide forte ½ tablet of 500 mg, and digoxin 0.25 mg ½ tablet
every second day due to atrial fibrillation and hypotension. The
patient was referred for multidetector computed tomography (MDCT) of
the chest, which was not performed later in the course.
At the second control visit on the fifth day of treatment (June 11,
2025), the patient had a total weight loss of 12 kg and minimal
residual edema (leg circumference: right 33 cm, 33 cm, 24 cm; left
34 cm, 33 cm, 24 cm). Lung examination showed normal breath sounds
with mildly reduced basal ventilation, no prolonged expiration, and
dullness to percussion at the bases below the 10th rib.
Echocardiographic evaluation demonstrated improvement in left
ventricular ejection fraction and improved diastolic function (left
ventricular compliance), with an E/e′ ratio of 7.5. Laboratory
results showed serum iron <1 μmol/L (normal 11–31) and ferritin 19.2
ng/mL (normal 20–250 ng/mL).
At the follow-up after two weeks (June 19, 2025), the patient
maintained excellent clinical improvement, with an additional 2 kg
weight loss (total 14 kg reduction from baseline), representing a
very good therapeutic response. He reported dizziness and
instability, attributed to hypotension (BP 90/55 mmHg and 80/50
mmHg), leading to dose reduction of hypotensive medications:
furosemide 500 mg ½ tablet every second day and sacubitril/valsartan
26/24 mg ¼ tablet twice daily. Due to lower leg pain, diosmin +
hesperidin 1000 mg once daily was added for venous symptoms.
New laboratory findings included: hemoglobin 88 g/L, erythrocyte
sedimentation rate (ESR) 55 mm/h, urea 15.4 mmol/L, creatinine 131
μmol/L, GFR 44.8 mL/min/1.73 m², and potassium 4.4 mmol/L.
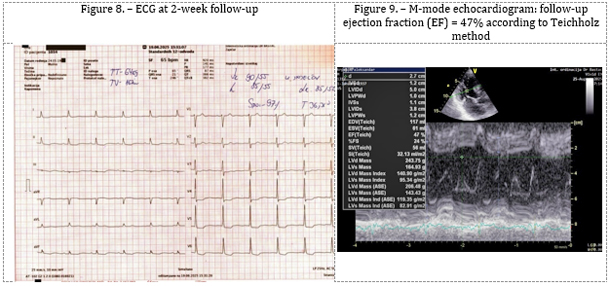
ECG: Atrial fibrillation with absolute arrhythmia, heart rate
65/min, ST depression up to 2 mm with negative T waves in V4–V6, and
ST depression up to 0.5 mm with negative T waves in leads I, II, and
aVL (Figure 8).
At routine follow-up after two months (August 25, 2025), the patient
was asymptomatic, with a further 2 kg weight reduction, no leg edema,
and no longer hypotensive. A marked increase in hemoglobin to 128
g/L was observed, attributed to iron therapy and correction of
hemodilution. Renal function normalized (GFR = 64 mL/min/1.73 m²).
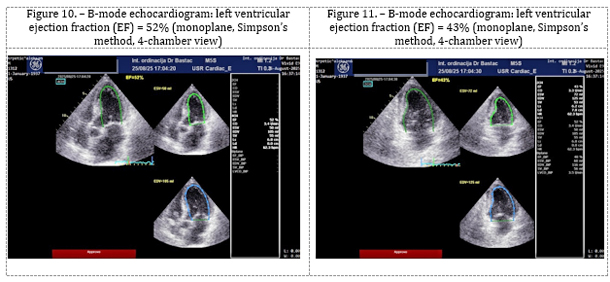
Echocardiography (Figures 9, 10, and 11) showed a significant
improvement in left ventricular systolic function, with M-mode EF of
47% and biplane Simpson EF of 46%, along with a reduction in left
ventricular dilation (LVEDD = 50 mm, LVESD = 40 mm) and resolution
of pulmonary hypertension (RVSP = 25 mmHg).
Further optimization of maintenance therapy was performed, including
reduction of furosemide dose to 500 mg ¼ tablet every second or
third day.
CLINICAL OUTCOME:
Following intensive intravenous diuretic therapy and three days
of day-hospital management with ECG monitoring and continuous
assessment of vital parameters, a significant clinical improvement
was achieved. The patient was subsequently transitioned to oral
therapy.
A total weight loss of 14 kg was recorded over a two-week period,
accompanied by complete resolution of peripheral edema and
normalization of lung auscultation findings. Pleural effusions
regressed to minimal levels. Left ventricular systolic function
remained preserved at approximately 46–47% ejection fraction, with
improvement in diastolic function on echocardiography.
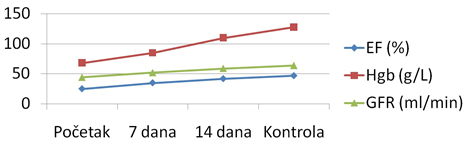
Laboratory parameters demonstrated progressive recovery of renal
function (GFR improved from 44 to 64 mL/min/1.73 m²) and a
significant increase in hemoglobin levels following iron
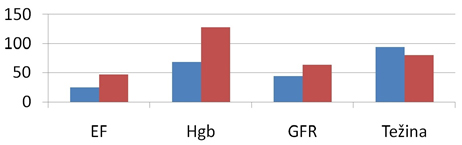
supplementation (Graph 1 and 2)
Graph 1. – Trend of changes in clinical parameters
during therapy
Graph 2. – Comparative presentation of pre- and
post-therapy effects
DISCUSSION
Anasarca represents an extreme form of fluid retention and a
marker of advanced heart failure with a poor prognosis [5].
Diuretics remain the cornerstone of congestion therapy, with
intravenous administration enabling faster and more effective
decongestion [6,13,14]. However, their use requires careful
monitoring due to the potential deterioration of renal function
[10,20], hypokalemia, and, less frequently, hypovolemia and
dehydration.
In this case, the improvement in renal function following therapy
suggests reversibility of cardiorenal syndrome after congestion
relief, as previously described in the literature [10,11].
Contemporary studies indicate that early initiation of SGLT2
inhibitors provides rapid clinical benefit and reduces
hospitalization rates [17,21–23]. ARNI therapy further contributes
to improved myocardial remodeling and reduced mortality [16].
Current guidelines emphasize the simultaneous or early sequential
implementation of the five foundational therapeutic pillars, which
is associated with the best clinical outcomes [7–9,24].
Congestion management remains the key therapeutic target, and
individualized intravenous diuretic therapy with careful monitoring
of body weight, urine output, and renal function is essential for
successful treatment [14,15]. Anemia is a common comorbidity in
heart failure, and its correction—particularly with intravenous iron
preparations—improves symptoms and quality of life [12,19].
This case highlights the importance of timely initiation of
intensive diuretic therapy, continuous monitoring of diuresis and
laboratory parameters, and an individualized approach depending on
comorbidities. It also demonstrates that an outpatient approach,
under adequate supervision, may be feasible in selected patients
with severe decompensated chronic heart failure that appears
terminal and refractory to treatment, although such patients are
most commonly managed in hospital settings [25].
CONCLUSION
Decompensated heart failure with anasarca represents a severe and
life-threatening condition requiring an aggressive yet carefully
titrated individualized therapeutic approach. Diuretic therapy
remains the cornerstone in controlling volume overload. This case
report highlights the importance of individualized diuretic therapy
combined with contemporary pharmacological strategies in patients
with the most severe forms of decompensated heart failure and
anasarca. Timely initiation of high-dose parenteral diuretics,
optimization of baseline therapy, and correction of associated
disorders led to significant clinical and laboratory improvement in
this patient.
This case also emphasizes the importance of intravenous therapy
administration under continuous ECG monitoring, along with close
observation and adjustment of treatment according to urine output,
blood pressure, heart rate, serum potassium and nitrogenous waste
levels, blood pressure, and oxygen saturation, in order to achieve
optimal outcomes. Particular importance is given to individualized
therapy and early recognition of refractoriness to standard oral
treatment strategies in chronic heart failure management.
The combination of intensive diuretic therapy and modern
pharmacological strategies can lead to substantial clinical
improvement even in patients with advanced disease, as demonstrated
in this case, which was successfully stabilized in an outpatient
day-hospital setting.
LITERATURE:
1. Savarese G, Lund LH. Global public health burden of heart
failure. Card Fail Rev. 2017;3(1):7–11.
2. Virani SS, Alonso A, Benjamin EJ, Bittencourt MS, Callaway CW,
Carson AP, et al. Heart disease and stroke statistics—2021 update: a
report from the American Heart Association. Circulation.
2021;143(8):e254–e743.
3. GBD 2022 Heart Failure Collaborators. Global, regional, and
national burden of heart failure, 1990–2022: a systematic analysis
for the Global Burden of Disease Study 2022. Lancet.
2022;400(10363):121–144.
4. Kasper DL, Fauci AS, Hauser SL, Longo DL, Jameson JL, Loscalzo J.
Harrison’s Principles of Internal Medicine. 19th ed. New York:
McGraw-Hill Education; 2015.
5. Eapen ZJ, Tang WHW, Felker GM, Hernandez AF. Defining true
clinical equipoise: cardiac cachexia versus anasarca in advanced
heart failure. Eur J Heart Fail. 2012;14(5):495–500.
6. Ponikowski P, Voors AA, Anker SD, Bueno H, Cleland JGF, Coats AJS,
et al. 2021 ESC Guidelines for the diagnosis and treatment of acute
and chronic heart failure. Eur Heart J. 2021;42(36):3599–3726.
7. Heidenreich PA, Bozkurt B, Aguilar D, Allen LA, Byun JJ, Colvin
MM, et al. 2023 ACC Expert Consensus Decision Pathway on Management
of Heart Failure with Reduced Ejection Fraction. J Am Coll Cardiol.
2023;81(18):1835–1878.
8. McDonagh TA, Metra M, Adamo M, Gardner RS, Baumbach A, Böhm M, et
al. 2023 Focused update of the 2021 ESC Guidelines for the diagnosis
and treatment of acute and chronic heart failure. Eur Heart J.
2023;44(37):3627–3739.
9. Greene SJ, Butler J, Fonarow GC. Simultaneous or rapid sequence
initiation of guideline-directed medical therapy for heart failure.
J Am Coll Cardiol. 2023;81(2):185–197
10. Damman K, Valente MAE, Voors AA, O’Connor CM, van Veldhuisen DJ,
Hillege HL. Renal impairment, worsening renal function, and outcome
in patients with heart failure: an updated meta-analysis. Eur Heart
J. 2014;35(7):455–469.
11. Ronco C, Haapio M, House AA, Anavekar N, Bellomo R. Cardiorenal
syndrome. J Am Coll Cardiol. 2008;52(19):1527–1539.
12. Ponikowski P, van Veldhuisen DJ, Comin-Colet J, Ertl G, Komajda
M, Mareev V, et al. Beneficial effects of long-term intravenous iron
therapy with ferric carboxymaltose in patients with symptomatic
heart failure and iron deficiency. Eur Heart J. 2015;36(11):657–668.
13. Felker GM, Lee KL, Bull DA, Redfield MM, Stevenson LW, Goldsmith
SR, et al. Diuretic strategies in patients with acute decompensated
heart failure. N Engl J Med. 2011;364(9):797–805.
14. Verbrugge FH, Mullens W, Tang WHW. Management of congestion in
heart failure: state-of-the-art review. Eur Heart J.
2023;44(24):2187–2200.
15. Damman K, Beusekamp JC, Boorsma EM, Swart HP, Smilde TDJ, Elvan
A, et al. Randomized, double-blind trial comparing high versus low
dose loop diuretics in acute heart failure. Eur J Heart Fail.
2023;25(3):456–466.
16. McMurray JJV, Packer M, Desai AS, Gong J, Lefkowitz MP, Rizkala
AR, et al. Angiotensin–neprilysin inhibition versus enalapril in
heart failure. N Engl J Med. 2014;371(11):993–1004.
17. McMurray JJV, Solomon SD, Inzucchi SE, Køber L, Kosiborod MN,
Martinez FA, et al. Dapagliflozin in patients with heart failure and
reduced ejection fraction. N Engl J Med. 2019;381(21):1995–2008.
18. Packer M, Anker SD, Butler J, Filippatos G, Pocock SJ, Carson P,
et al. Cardiovascular and renal outcomes with empagliflozin in heart
failure. N Engl J Med. 2020;383(15):1413–1424.
19. Anker SD, Comin Colet J, Filippatos G, Willenheimer R, Dickstein
K, Drexler H, et al. Ferric carboxymaltose in patients with heart
failure and iron deficiency. N Engl J Med. 2009;361(25):2436–2448.
20. Mullens W, Damman K, Harjola VP, Mebazaa A, Brunner-La Rocca HP,
Martens P, et al. The use of diuretics in heart failure with
congestion – a position statement from the Heart Failure Association
of the European Society of Cardiology. Eur J Heart Fail.
2019;21(2):137–155.
21. Butler J, Anker SD, Filippatos G, Khan MS, Ferreira JP, Pocock
SJ, et al. Empagliflozin and outcomes in patients with heart failure
with preserved, mildly reduced, and reduced ejection fraction:
pooled analysis of EMPEROR trials. Eur Heart J. 2023;44(5):449–460.
22. Vaduganathan M, Claggett BL, Jhund PS, Cunningham JW, Ferreira
JP, Zannad F, et al. Time to clinical benefit of dapagliflozin in
patients with heart failure with reduced ejection fraction: insights
from the DAPA-HF trial. Circulation. 2023;147(5):375–385.
23. Docherty KF, Jhund PS, Inzucchi SE, Køber L, Kosiborod MN,
Martinez FA, et al. Effects of dapagliflozin across the spectrum of
ejection fraction in heart failure. Nat Med. 2024;30(1):123–131.
24. Maddox TM, Januzzi JL Jr, Allen LA, Breathett K, Butler J, Davis
LL, et al. 2024 ACC Expert Consensus Decision Pathway for
optimization of heart failure treatment. J Am Coll Cardiol.
2024;83(9):1056–1098.
25. Ambrosy AP, Fonarow GC, Butler J, Chioncel O, Greene SJ,
Vaduganathan M, et al. The global health and economic burden of
hospitalizations for heart failure: lessons learned from
hospitalized heart failure registries. J Am Coll Cardiol.
2014;63(12):1123–1133.
26. Zannad F, Ferreira JP, Pocock SJ, Anker SD, Butler J, Filippatos
G, et al. SGLT2 inhibitors in patients with heart failure with
reduced ejection fraction: a meta-analysis of the EMPEROR-Reduced
and DAPA-HF trials. Lancet. 2020;396(10244):819–829.
27. Anker SD, Butler J, Filippatos G, Ferreira JP, Bocchi E, Böhm M,
et al. Semaglutide in patients with heart failure with preserved
ejection fraction and obesity (STEP-HFpEF trial). N Engl J Med.
2023;389(12):1069–1084.
|
|
|
|









