| |
|
|
INTRODUCTION
Intermittent fasting or fasting as a therapeutic method has been
used at least since the 5th century BC. At that time, Hippocrates
recommended abstinence from food or drink for patients exhibiting
certain disease symptoms. Some doctors later recognized the instinct
of fasting (in patients who, in certain diseased states, naturally
experience a loss of appetite) and believed that providing food
during such conditions was unnecessary and possibly even harmful,
believing that fasting was an important natural part of the recovery
process. The understanding of the physiological effects of fasting
began to develop in the second half of the 19th century when some of
the first organized fasting studies were conducted on animals and
humans. In the 20th century, as knowledge of nutrition and the
nutritional needs of the human body grew, fasting methods became
more sophisticated, and a wide range of ways to apply this form of
eating emerged.
The term fasting for Orthodox Christians refers to the abstention
from certain types of food, primarily meat, dairy products, and
eggs, and in some fasting periods, even fish, oil, and alcoholic
beverages are avoided. In this paper, fasting will refer to the
occasional interruption of the intake of any type of food (or the
consumption of food and caloric drinks in minimal quantities) during
periods typically ranging from 12-36 hours. Intermittent fasting
(IF) can be practiced daily, alternating every other day, twice a
week, or once a week. Fasting can be practiced for religious reasons
as well as for health purposes. Members of certain religious
communities traditionally fast on specific days of the week or
calendar year. In many healthcare institutions, patients under
medical supervision follow a fasting regimen or calorie restriction
to control body weight, prevent, or treat diseases.
Fasting differs from caloric restriction (CR), where daily caloric
intake is chronically reduced by 20-40%, but meal frequency is
maintained. Unlike fasting and CR, starvation is chronic nutritional
insufficiency often used as a substitute for the word fasting, but
it is also used to define extreme forms of fasting (e.g.,
starvation), which can lead to degeneration and death. Research on
animal models, as well as studies on humans, shows that fasting
leads to ketogenesis, promotes strong changes in metabolic pathways
and cellular processes such as stress resistance, lipolysis, and
autophagy, and can have medical applications [1].
Intermittent fasting is technically not just a diet plan but a way
of eating that focuses on timing rather than the type of food.
Studies on animals and humans have shown that many health benefits
of intermittent fasting are not solely a result of reduced free
radical production or weight loss. Instead, intermittent fasting
triggers evolutionarily preserved, adaptive cellular responses that
improve glucose regulation, increase stress resistance, and suppress
inflammation. During fasting, cells activate pathways that enhance
defense against oxidative and metabolic stress and those that remove
or repair damaged molecules [2]. The remarkable effects of typical
CR (20-40%) on aging and diseases in mice and rats are often seen as
mammalian responses during evolution to adapt to periods of limited
food availability. However, the cellular and molecular mechanisms
responsible for the protective effects of CR likely evolved billions
of years earlier in prokaryotes attempting to survive in
environments that were largely or completely devoid of energy
sources [3]. For example, the bacterium E. coli, transferred from a
nutrient-rich medium to a calorie-free medium, survives four times
longer, an effect reversed by adding various nutrients, but not
acetate, a carbon source associated with starvation conditions [4].
The shortening of the bacterium's lifespan in a rich medium, but not
acetate, suggests that a ketone-body-like carbon source such as
acetate could be part of an "alternative metabolic program" that
evolved over billions of years in microorganisms, now allowing
mammals to survive periods of food scarcity by obtaining most of
their energy through fatty acid and ketone body catabolism,
including acetoacetate and β-hydroxybutyrate [5]. In Saccharomyces
cerevisiae (brewer's yeast), transferring cells from a standard
growth medium to water also causes consistent double chronological
lifespan extension, as well as a significant increase in resistance
to multiple stressors [6]. Another organism model where fasting
extends lifespan is the nematode Caenorhabditis elegans. Food
deprivation conditions achieved by feeding the worms with little or
no bacteria lead to significant lifespan extension [1]. In the fruit
fly, most studies suggest that intermittent food deprivation does
not affect lifespan. However, it has consistently been shown that
reducing or diluting food extends the longevity of Drosophila,
suggesting that flies may benefit from dietary restriction but may
be sensitive even to short periods of starvation. Taken together,
these results indicate that food deprivation can lead to
lifespan-extending effects across a wide range of organisms but also
emphasize that different organisms have different responses to
fasting [1].
Metabolic Changes During Fasting
In most mammals, the liver serves as the main reservoir for glucose,
which is stored in the form of glycogen. In humans, depending on the
level of physical activity, after 12 to 24 hours of fasting, the
glucose levels in the serum drop by 20% or more. The glycogen
reserves in the liver become depleted. The body shifts to a
metabolic state in which the liver and kidneys produce glucose from
non-carbohydrate sources, such as glycogenic amino acids from
muscles (isoleucine, phenylalanine, tyrosine, tryptophan), glycerol
from fats, and lactic acid, followed by lipolysis in adipose tissue,
releasing free fatty acids and glycerol, which the body uses as
energy. While most tissues can use fatty acids for energy, during
extended fasting periods, the brain, in addition to glucose, relies
on ketone bodies β-hydroxybutyrate and acetoacetate for energy
consumption. Ketone bodies are produced in hepatocytes from acetyl-CoA
formed through β-oxidation of fatty acids released into the
bloodstream by adipocytes, and also through the conversion of
ketogenic amino acids (leucine and lysine). After 3-5 days of
fasting, the liver produces ketone bodies (beta-hydroxybutyrate and
acetoacetate) from fatty acids through ketogenesis, which become the
main energy source for the brain and muscles. At the same time, the
use of protein as an energy source decreases. After 5 days without
food, the brain almost completely switches to ketone bodies as an
energy source, thus protecting muscle mass. Minimal gluconeogenesis
still occurs, approximately 80 grams daily, with glucose being
produced only in amounts necessary for cells that cannot use ketone
bodies (e.g., erythrocytes and some parts of the brain) [7].
Depending on body weight and composition, ketone bodies, free fatty
acids, and gluconeogenesis allow most people to survive for 30 or
more days in conditions of food scarcity, and allow certain species,
such as royal penguins, to survive without food for more than 5
months [8].
Metabolic Adaptations to Intermittent Fasting
In humans, the three most studied intermittent fasting regimens
are alternate-day fasting (one day without food, the next day food
ad libitum), 5:2 intermittent fasting (fasting for 2 days each
week), and daily time-restricted feeding. Diets that significantly
reduce caloric intake for 1 day or more each week (e.g., reducing to
500–700 calories per day) lead to increased levels of ketone bodies
on those days [9, 10].
The metabolic shift from using glucose as a fuel source to using
fatty acids and ketone bodies ("metabolic switch") results in a
decreased respiratory quotient (the ratio of carbon dioxide produced
to oxygen consumed), indicating greater metabolic flexibility and
efficiency in energy production from fatty acids and ketone bodies
[11].
Ketone bodies are not only fuels used during fasting periods; they
are powerful signaling molecules with significant effects on
cellular and organ functions. Ketone bodies act as metabolic signals
that regulate epigenetics through beta-hydroxybutyrate (BHB), which
inhibits histone deacetylase (HDAC). This inhibition then results in
an antioxidant response and lifespan extension. They increase the
activity of sirtuins (especially SIRT1 and SIRT3), which reduces
oxidative stress. Ketones also modulate inflammatory and antioxidant
pathways by promoting the activation of Nrf2 (Nuclear factor
erythroid 2-related factor 2), the primary regulator of the
antioxidant response, and by reducing the activity of NF-κB (Nuclear
Factor kappa-light-chain-enhancer of activated B cells), a key
factor in inflammatory processes. Additionally, ketones increase
stress resistance and promote autophagy through AMPK (AMP-activated
protein kinase), which stimulates autophagy and mitochondrial
biogenesis, contributing to cellular health and stress resilience.
Ketone bodies indirectly inhibit mTORC1 activity (the
mechanistic/mammalian target of rapamycin complex), the major
regulator of cellular growth and protein synthesis. Mechanistically,
mTOR is a key regulator of autophagy and cellular metabolism in
mammals. Reduced mTOR activity shifts cellular resources from
non-essential anabolic reactions toward catabolic processes,
including activation of complexes important for autophagy. Reduced
mTOR activity is linked to extended lifespan and protection from
age-related diseases [12].
By affecting these key cellular pathways, ketone bodies produced
during fasting have profound effects on systemic metabolism.
Furthermore, ketone bodies stimulate the expression of genes for
brain-derived neurotrophic factor, with implications for brain
health and psychiatric and neurodegenerative disorders [13]. Many
studies have shown that some of the benefits of intermittent fasting
are separate from its effects on weight loss. These benefits
include: improvements in glucose regulation, regulation of blood
pressure, reduction in heart rate, increased endurance training
efficiency, and loss of abdominal fat [14].].
Picture 1. POSSIBLE MECHANISMS OF INTERMITTENT
FASTING ON HEALTH IMPROVEMENT
Source (25.02.2025): Song DK, Kim YW. Beneficial effects of
intermittent fasting: a narrative review. J Yeungnam Med Sci. 2023
Jan;40(1):4-11. https://pmc.ncbi.nlm.nih.gov/articles/PMC9946909/
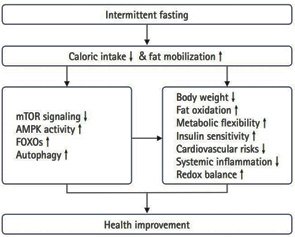
The Effects of Intermittent Fasting on Health and Aging
After nearly a century of research on caloric restriction in
animal models, the general conclusion was that reduced food intake
significantly extends the lifespan of the animals studied. In one of
the earliest studies on intermittent fasting, Goodrick and
colleagues showed that the average lifespan of rats was extended by
up to 80% when maintained on an alternate day feeding regimen,
starting when they were young adults. However, the magnitude of the
effects of caloric restriction on healthspan and lifespan varies and
can be influenced by gender, diet, age, and genetic factors [2]. A
meta-analysis of data available from 1934 to 2012 showed that
caloric restriction in rats extended the average lifespan by 14 to
45%, but only by 4 to 27% in mice [15].
Conflicting results from two significant studies on monkeys raised
doubts about the relationship between improved health status and
extended lifespan through caloric restriction. One study on rhesus
monkeys showed a positive effect of caloric restriction on both
health and survival [16], while another study, also on rhesus
monkeys, did not show a significant reduction in mortality with
caloric restriction, despite clear improvements in overall health
[17]. A subsequent study showed that differences in daily caloric
intake, the timing of the intervention, food composition, feeding
protocols, gender, and genetic background could explain the varying
effects of caloric restriction on lifespan in the two previous
studies [18].
Intermittent fasting in humans alleviates obesity, insulin
resistance, dyslipidemia, hypertension, and inflammation. It seems
that intermittent fasting provides more health benefits than can be
attributed solely to a reduction in caloric intake. In one study, 16
healthy participants who underwent an alternate-day fasting regimen
for 22 days lost 2.5% of their initial body weight and 4% of body
fat, along with a 57% reduction in fasting insulin levels [19]. In
two other studies, approximately 100 overweight women in each study
were divided into two groups: one following a 5:2 intermittent
fasting regimen, and the other reducing their daily caloric intake
by 25%. Participants in both groups lost the same amount of weight
during the 6-month period, but those in the 5:2 intermittent fasting
group had a greater increase in insulin sensitivity and a larger
reduction in waist circumference [20].
Benefits of Intermittent Fasting for the Aged and Diseased
Vasculature
Vascular aging involves arterial stiffness and the formation of
fibrolipid lesions in the arterial wall, leading to atherosclerosis.
The main clinical manifestations of atherosclerosis include coronary
artery disease, ischemic stroke, and peripheral artery disease,
which are caused by hyperlipoproteinemia (low-density lipoprotein
LDL cholesterol), inflammation, vascular remodeling, and plaque
formation [21]. Intermittent fasting (IF) is associated with a
reduction in LDL cholesterol levels [22]. In rats, both in the
absence and presence of various stressors (e.g., swimming),
intermittent fasting reduces resting blood pressure and heart rate.
The reduction in blood pressure may be partially due to enhanced
endothelial cell-dependent vasodilation [23,24].
Furthermore, intermittent fasting activates the parasympathetic
nervous system by stimulating brain cells. During fasting,
neurotrophic factors are released, and acetylcholine is secreted,
which, via the vagus nerve, leads to a decrease in heart rate and
blood pressure [25].
Risks of Intermittent Fasting for the Aged and Diseased Heart
and Vasculature
Despite the numerous health benefits of intermittent fasting
(IF), some risks have been reported in various models of
cardiovascular diseases. For example, rats subjected to
alternate-day fasting for 6 months showed reduced diastolic
compliance of the left ventricle and diminished cardiac reserve
[26].
The efficacy of activating autophagy in senescent cells remains a
subject of ongoing debate, as autophagy has also been reported to
promote cellular aging by facilitating the synthesis of
aging-associated proteins. Furthermore, excessive stimulation of
autophagy can lead to several pathological outcomes, including
inhibition of angiogenesis [27]. Therefore, reduced regenerative
capacity of the endothelium and the accumulation of senescent cells
in older individuals may potentially limit the beneficial effects of
intermittent fasting on vascular health.
In older adults, particularly those with hypertension or other
cardiovascular diseases, potential fluctuations in blood pressure
during intermittent fasting periods may raise concerns regarding
cardiovascular risks, orthostasis, and fall-related injuries [28].
Another risk associated with intermittent fasting arises from
metabolic changes, such as electrolyte imbalances or activation of
the sympathetic nervous system, which can predispose older
individuals to dehydration and cardiac arrhythmias, especially in
the presence of pre-existing heart conditions.
In summary, due to the lack of data, the implementation of
intermittent fasting in older individuals or patients with
cardiovascular diseases requires careful consideration due to
potential risks, which depend on the specific fasting regimen,
cardiac condition, comorbidities, gender, and age [29].
Effect of Intermittent Fasting on Cardiometabolic Health
The weight loss induced by intermittent fasting is primarily
attributed to a reduction in fat mass. Studies have documented
reductions in subcutaneous and visceral fat, with the latter being
particularly beneficial due to its association with metabolic
dysregulation and increased cardiovascular risk [30].
Waist circumference, a key anthropometric marker of cardiovascular
risk used to assess abdominal adiposity, is strongly associated with
all-cause mortality and cardiovascular mortality. It has been shown
that waist circumference significantly decreases in individuals
practicing intermittent fasting. This reduction in waist
circumference is directly correlated with a lower risk of coronary
artery disease and other cardiovascular pathologies [31].
Moreover, intermittent fasting improves several cardiovascular risk
factors, including blood pressure, lipid profile, resting pulse,
glucose and insulin levels, and insulin resistance. Furthermore,
intermittent fasting can alleviate chronic inflammation associated
with aging by reducing systemic inflammatory markers and oxidative
stress linked to atherosclerosis in humans [29].
Aging is a critical factor in the pathogenesis and progression of
heart failure (HF), increasing the incidence and severity of HF.
Several studies have suggested that intermittent fasting may improve
risk factors associated with the development of HF in both healthy
individuals and those with obesity and ischemic heart disease [32].
For instance, in one study, participants who reported routinely
practicing intermittent fasting at least once a month for a minimum
of 5 years experienced a 71% reduction in the risk of heart failure
compared to those who did not fast [33]. Another prospective
observational study found that later periods of the first and last
meals were associated with a greater risk of cardiovascular events,
consistent with randomized studies reporting that late evening meals
may exacerbate cardiovascular risk factors [34]. These inconsistent
findings require further research into the relationship between
intermittent fasting and heart failure through large randomized
controlled trials investigating the effect of fasting at different
times of the day.
The effect of intermittent fasting on muscle health is a topic of
debate as it remains unclear whether intermittent fasting preserves
lean muscle mass during weight loss or exacerbates the breakdown of
muscle proteins and net catabolism. While some earlier studies
suggested a reduction in lean mass with dieting, the general
consensus is that intermittent fasting does not have a detrimental
effect on lean mass, even with weight loss in otherwise healthy
individuals, as well as in insulin-treated diabetic patients [35].
Physical and Cognitive Effects of Intermittent Fasting
In both animals and humans, physical function improves with
intermittent fasting. For example, despite having similar body
weights, mice maintained on an intermittent fasting regimen exhibit
better running endurance than mice with unlimited access to food
[2]. Balance and coordination are also improved in animals following
time-restricted daily feeding or alternate-day fasting regimens
[36]. Juveniles who fast for 16 hours daily lose fat while
maintaining muscle mass over 2 months of intense training [37].
Animal studies show that intermittent fasting improves cognition
across various domains, including spatial memory, associative
memory, and working memory [38]. Alternate-day fasting and daily
caloric restriction counteract the harmful effects of obesity,
diabetes, and neuroinflammation on spatial learning and memory. In a
clinical trial, older adults on a short-term caloric restriction
regimen experienced improved verbal memory. In a study involving
overweight adults with mild cognitive impairment, 12 months of
caloric restriction led to improvements in verbal memory, executive
function, and global cognition [39, 40]. A large, multicenter,
randomized clinical trial demonstrated that 2 years of daily caloric
restriction resulted in significant improvements in working memory
[41]. Further research is needed to explore the relationship between
intermittent fasting and cognition in older adults, especially
considering the absence of pharmacological therapies affecting brain
aging and the progression of neurodegenerative diseases.
DISCUSSION
An increasing body of evidence supports intermittent fasting (IF)
in all its variations as a potentially safe and feasible dietary
intervention for improving human health. IF can improve
physiological and molecular markers of aging and provide benefits
for cardiovascular and metabolic health in patients with obesity,
type 2 diabetes, metabolic syndrome, and heart failure [29].
Significant weight loss and other health benefits have been
associated with two types of intermittent fasting: modified
alternate-day fasting (alternating between a day of normal food
intake and a day consuming up to 600 calories) and the "5:2 diet" (2
days of no caloric intake per week) [42].
While clinical evidence is mostly of a research nature, these
studies provide a solid rationale for investigating the efficacy of
IF in improving cardiovascular health, particularly in the elderly
population at risk for or already experiencing cardiovascular
diseases. Future randomized trials with larger sample sizes and
longer durations will be necessary to assess the long-term outcomes,
adherence, and safety of IF, especially in older participants.
Despite the health benefits of intermittent fasting and its
applicability to many diseases, there are barriers to widespread
adoption of these dietary patterns in the community and by patients.
First, the traditional three-meal-a-day pattern with snacks is so
ingrained in our culture that patients or physicians rarely consider
changing this eating pattern. The abundance of food and extensive
marketing in developed countries also pose significant barriers that
must be overcome. Second, when transitioning to an intermittent
fasting regimen, many people experience hunger, irritability, and
reduced concentration during periods of food restriction. However,
these initial side effects usually subside within a month, and
patients should be informed of this [2].
Given the limitations and risks outlined above, patients should be
cautioned that eating patterns involving extended periods without
food could pose risks for people with diabetes who are on insulin or
otherwise prone to hypoglycemia [42]. Physician education is also
recommended for patients with a range of chronic conditions or at
risk of such conditions, particularly those associated with
overeating and a sedentary lifestyle, on how to implement
intermittent fasting for prevention or as part of early treatment
for these conditions.
Another important aspect to consider is that intermittent fasting
alters the gastrointestinal microbiome [43]. Fasting regimens appear
to have a positive impact on gut microbiota. Future studies
characterizing the health effects of fasting regimens on the human
microbiome have the potential to make an important contribution to
this field. Therefore, it will also be crucial to investigate
changes in metabolites produced by gut bacteria, focusing on the
molecular mechanisms underlying the effects of intermittent fasting
on cellular aging.
CONCLUSION
Intermittent fasting relies on the concept of a "metabolic
switch," which involves a shift from glucose-dependent metabolism
during a typical diet to ketones derived from fat cells during
fasting. This "metabolic switch" may improve glucose regulation and
reduce inflammation. The stress of fasting also increases autophagy,
which removes damaged molecules. Given these physiological changes,
intermittent fasting can offer significant long-term health
benefits. Animal models of intermittent fasting show that this
dietary pattern improves the health of the animals throughout their
lifespan. Clinical studies in human models have also demonstrated
significant health benefits, although these studies have mostly
involved relatively short-term interventions lasting several months.
Preclinical studies and clinical trials have shown that intermittent
fasting offers a broad range of advantages for many health
conditions, such as obesity, diabetes, cardiovascular diseases,
certain cancers, and neurological disorders. Numerous studies
suggest that intermittent fasting regimens may be a promising
approach for weight loss and improving metabolic health in people
who can tolerate periods without food or consume very little at
certain times of the day or on specific days of the week. For
healthy, normal-weight, or obese adults, there is little evidence
that intermittent fasting regimens are harmful physically or
mentally. Future studies should determine whether the benefits
observed in animal models can be sustained over long-term
intermittent fasting in humans of various ages and health statuses.
Further understanding of the processes linking intermittent fasting
to many health benefits may allow us to develop targeted
pharmacological therapies, including interventions on the gut
microbiome, that mimic the effects of intermittent fasting without
requiring fundamental changes to eating habits.
LITERATURE:
- Longo VD, Mattson MP. Fasting: molecular mechanisms and
clinical applications. Cell Metab. 2014;19(2):181-92. doi:
10.1016/j.cmet.2013.12.008. Epub 2014 Jan 16. PMID: 24440038;
PMCID: PMC3946160.
- de Cabo R, Mattson MP. Effects of Intermittent Fasting on
Health, Aging, and Disease. N Engl J Med.
2019;381(26):2541-2551.
- Fontana L, Partridge L, Longo VD. Extending healthy lPPe
span--from yeast to humans. Science. 2010;328:321–326. doi:
10.1126/science.1172539.
- Gonidakis S, Finkel SE, Longo VD. Genome-wide screen
identPPies Escherichia coli TCA-cycle-related mutants with
extended chronological lPPespan dependent on acetate metabolism
and the hypoxia-inducible transcription factor ArcA. Aging Cell.
2010;9:868–881. doi: 10.1111/j.1474-9726.2010.00618.x.
- Cahill GF., Jr Fuel metabolism in starvation. Annu Rev Nutr.
2006;26:1–22. doi: 1146/annurev.nutr.26.061505.111258.
- Longo VD, Shadel GS, Kaeberlein M, Kennedy B. Replicative
and chronological aging in Saccharomyces cerevisiae. Cell Metab.
2012;16:18–31. doi: 10.1016/j.cmet.2012.06.002.
- Sander Kersten The impact of fasting on adipose tissue
metabolism Biochimica et Biophysica Acta (BBA) - Molecular and
Cell Biology of Lipids, 2023;1868(3):159262. ISSN 1388-1981.
- Eichhorn G, Groscolas R, Le Glaunec G, Parisel C, Arnold L,
Medina P, Handrich Y. Heterothermy in growing king penguins. Nat
Commun. 2011;2:435. doi: 10.1038/ncomms1436.
- Skrha J, Kunesová M, Hilgertová J,Weiserová H, Krízová J,
Kotrlíková E. Short term very low calorie diet reduces oxidative
stress in obese type 2 diabetic patients. Physiol Res
2005;54:33-9.
- Harvie MN, Pegington M, Mattson MP, et al. The effects of
intermittent or continuous energy restriction on weight loss and
metabolic disease risk markers: a randomized trial in young
overweight women. Int J Obes (Lond) 2011;35:714-27.
- Di Francesco A, Di Germanio C, Bernier M, de Cabo R. A time
to fast. Science. 2018;362(6416):770-775.
- Hwang, C.Y.; Choe, W.; Yoon, K.-S.; Ha, J.; Kim, S.S.; Yeo,
E.-J.; Kang, I. Molecular Mechanisms for Ketone Body Metabolism,
Signaling Functions, and Therapeutic Potential in Cancer.
Nutrients 2022;14:4932.
- Mattson MP, Moehl K, Ghena N,Schmaedick M, Cheng A.
Intermittent metabolic switching, neuroplasticity and brain
health. Nat Rev Neurosci 2018;19:63-80.
- Anson RM, Guo Z, de Cabo R, et al .Intermittent fasting
dissociates beneficial effects of dietary restriction on glucose
metabolism and neuronal resistance to injury from calorie
intake. Proc Natl Acad Sci U S A 2003;100:6216-20.
- Swindell WR. Dietary restriction in rats and mice: a
meta-analysis and review of the evidence for genotype-dependent
effects on lPPespan. Ageing Res Rev 2012;11:254-70.
- Colman RJ, Anderson RM, Johnson SC, et al. Caloric
restriction delays diseaseonset and mortality in rhesus monkeys
Science 2009;325:201-4.
- Mattison JA, Roth GS, Beasley TM, et al. Impact of caloric
restriction on health and survival in rhesus monkeys from the
NIA study. Nature 2012;489:318-21.
- Mattison JA, Colman RJ, Beasley TM,et al. Caloric
restriction improves health and survival of rhesus monkeys. Nat
Commun 2017;8:14063.
- Heilbronn LK, Smith SR, Martin CK,Anton SD, Ravussin E.
Alternate-day fasting in nonobese subjects: effects on body
weight, body composition, and energy metabolism. Am J Clin Nutr
2005;81:69-73.
- Harvie M, Wright C, Pegington M, et al.The effect of
intermittent energy and carbohydrate restriction v. daily energy
restriction on weight loss and metabolic disease risk markers in
overweight women. Br J Nutr 2013;110:1534-47.
- Herrington W., Lacey B., Sherliker P. Epidemiology of
atherosclerosis and the potential to reduce the global burden of
atherothrombotic disease Circ Res. 2016;18:535-546.
- Chen Y., Su J., Yan Y. Intermittent fasting inhibits
high-fat diet-induced atherosclerosis by ameliorating
hypercholesterolemia and reducing monocyte chemoattraction Front
Pharmacol. 2021;12:719750.
- Wan R., Camandola S., Mattson M.P. Intermittent food
deprivation improves cardiovascular and neuroendocrine responses
to stress in rats. J Nutr. 2003;133:1921-1929.
- Razzak R.L., Abu-HozaPPa B.M., Bamosa A.O. Assessment of
enhanced endothelium-dependent vasodilation by intermittent
fasting in Wistar albino rats Indian J Physiol Pharmacol.
2011;55:336-342.
- Wan R., Weigand L.A., Bateman R. Evidence that BDNF
regulates heart rate by a mechanism involving increased
brainstem parasympathetic neuron excitability J Neurochem.
2014;129:573-580.
- Ahmet I., Wan R., Mattson M.P. Chronic alternate-day fasting
results in reduced diastolic compliance and diminished systolic
reserve in rats J Card Fail. 2010;16:843-853.
- Shabkhizan R., Haiaty S., Moslehian M.S. The beneficial and
adverse effects of autophagic response to caloric restriction
and fasting Adv Nutr. 2023;14:1211-1225.
- Bencivenga L., De Souto Barreto P., Rolland Y. Blood
pressure variability: a potential marker of aging Ageing Res
Rev. 2022;80:101677.
- Mualla O. et al. Risks and Benefits of Intermittent Fasting
for the Aging Cardiovascular System Canadian Journal of
Cardiology, 2024;40(8):1445-57.
- Heilbronn L.K., Smith S.R., Martin C.K. Alternate-day
fasting in nonobese subjects: effects on body weight, body
composition, and energy metabolism Am J Clin Nutr.
2005;81:69-73.
- Hoddy, K.K. Kroeger, C.M. Trepanowski, J.F. Meal timing
during alternate day fasting: Impact on body weight and
cardiovascular disease risk in obese adults Obesity (Silver
Spring). 2014;22:2524-2531.
- Sedej, S. AbdellatPP, M.Metabolic therapy for managing heart
failure with preserved ejection fraction J Mol Cell Cardiol.
2022;168:68-69.
- Bartholomew, C.L. Muhlestein, J.B. Anderson, J.L.
Association of periodic fasting lPPestyles with survival and
incident major adverse cardiovascular events in patients
undergoing cardiac catheterization Eur J Prev Cardiol.
2022;28:1774-1781.
- Palomar-Cros, A. ∙ Andreeva, V.A. ∙ Fezeu, L.K. Dietary
circadian rhythms and cardiovascular disease risk in the
prospective NutriNet-Santé cohort Nat Commun. 2023;14:7899.
- Obermayer, A. ∙ Tripolt, N.J. ∙ Pferschy, P.N. Efficacy and
safety of Intermittent Fasting in People With Insulin-Treated
Type 2 Diabetes (INTERFAST-2)-a randomized controlled trial
Diabetes Care. 2023;46:463-468.
- Chaix A, Zarrinpar A, Miu P, Panda S. Time-restricted
feeding is a preventative and therapeutic intervention against
diverse nutritional challenges. Cell Metab 2014;20:991-1005.
- Moro T, Tinsley G, Bianco A, et al. Effects of eight weeks
of time-restricted feeding (16/8) on basal metabolism, maximal
strength, body composition, inflammation, and cardiovascular
risk factors in resistance-trained males. J Transl Med
2016;14:290.
- Wahl D, Coogan SC, Solon-Biet SM, et al. Cognitive and
behavioral evaluation of nutritional interventions in rodent
models of brain aging and dementia. Clin Interv Aging
2017;12:1419-28.
- Witte AV, Fobker M, Gellner R, Knecht S, Flöel A. Caloric
restriction improves memory in elderly humans. Proc Natl Acad
Sci U S A 2009;106:1255-60.
- Horie NC, Serrao VT, Simon SS, et al. Cognitive effects of
intentional weight loss in elderly obese individuals with mild
cognitive impairment. J Clin Endocrinol Metab 2016;101:1104-12.
- Wan R, Camandola S, Mattson MP. Intermittent food
deprivation improves cardiovascular and neuroendocrine responses
to stress in rats. J Nutr 2003;133: 1921-9.
- Vega C.P. Is Intermittent Fasting Beneficial? https://www.medscape.org/viewarticle/967107
(preuzeto 22.11.2024)
- MaPPeld A., Bartolomaeus H., Löber U. et al. Fasting alters
the gut microbiome reducing blood pressure and body weight in
metabolic syndrome patients Nat Commun. 2021;12:1970.
|
|
|
|

