| |
|
|
INTRODUCTION Colorectal cancer (CRC) is one of the most
common gastrointestinal malignancies, with significant morbidity and
mortality worldwide. Epidemiological data indicate that the
incidence of CRC is increasing, which can be attributed to factors
such as population aging, changes in dietary habits, and the rising
prevalence of obesity. Surgical treatment remains the primary
therapeutic option for most patients; however, nutritional status
plays a crucial role in treatment outcomes, postoperative recovery,
and patients’ quality of life.
To understand the importance of nutritional support in surgical
patients, it is necessary to know what happens to metabolism during
and after surgery. Surgical intervention, as well as trauma, leads
to the release of mediators of the systemic inflammatory response,
resulting in the catabolism of glycogen, proteins, and fats, with
subsequent release of glucose, free fatty acids, and amino acids
into the circulation. The result is a loss of muscle mass, which
impairs functional recovery after surgery. Preservation of muscle
stores, reduction of lipolysis, and glucose oxidation are desirable
factors in the postoperative period. Perioperative nutritional
support alone has limited impact in the immediate postoperative
period, within the first few hours after surgery, when the body is
in a catabolic state. Nutritional support and physical activity are
essential for the restoration of peripheral muscle mass and
functional recovery after major surgery. Optimization of nutrition
in moderately malnourished patients should be considered over a
period of 7–10 days. In severely malnourished patients, the focus of
intervention should be on correcting hypoglycemia, dehydration,
electrolyte imbalance, infections, and micronutrient deficiencies.
[1].
Definitive surgical treatment (extensive dissections, high-risk
anastomoses) is recommended at a later stage, once the infectious
focus has been resolved. Malnutrition is common in patients with
colorectal cancer, as a result of chronic inflammation due to
malignant disease, impaired food intake, renal dysfunction, and
liver failure, and it may increase the risk of postoperative
complications and prolong hospitalization. The causes of
malnutrition in these patients are multifactorial and include
reduced food intake due to gastrointestinal symptoms (nausea,
vomiting, diarrhea, constipation), metabolic changes associated with
malignancy, as well as the consequences of surgical and oncological
treatments. Nutritional therapy (support) may also be indicated in
patients without obvious, clinically apparent malnutrition as a
consequence of the underlying oncological disease, in cases where
prolonged interruption or reduction of oral intake is expected [2].
The European Society for Clinical Nutrition and Metabolism (ESPEN)
recommends systematic assessment of nutritional status and
implementation of targeted nutritional interventions to improve
treatment outcomes. Enteral and parenteral nutrition, oral
nutritional supplements (ONS), and individualized nutritional
approaches can significantly improve nutritional status and reduce
postoperative complications. [3].
Preoperative nutritional support may reduce postoperative infections
and improve wound healing, while an adequate perioperative
nutritional strategy can contribute to faster recovery and a
reduction in hospital treatment costs. [2].
ESPEN has redefined the criteria for the assessment of malnutrition
based on two criteria:
BMI < 18.5 kg/m².
Total body weight loss > 10% or > 5% within the last three months,
together with a reduced BMI. [2].
Disease-related malnutrition (DRM) is a subcategory according to the
WHO, characterized by malnutrition with a BMI < 18.5 kg/m². The
assessment of metabolic risk in DRM can be easily identified using
NRS-2002. Recent studies have shown that preoperative albumin levels
and body weight loss have a significant impact on postoperative
outcomes and complication rates. [4].
Assessment of nutritional status in patients undergoing colorectal
cancer surgery requires a multidisciplinary approach, including
anthropometric, laboratory, and clinical parameters. The use of
validated screening tools, such as the Nutritional Risk Screening
(NRS-2002) and the Subjective Global Assessment (SGA), enables early
identification of patients at increased nutritional risk and timely
intervention. In addition, biochemical markers such as albumin,
prealbumin, and C-reactive protein (CRP) play an important role in
assessing nutritional status and predicting postoperative outcomes.
[5].
The modified Glasgow Prognostic Score (mGPS), as an indicator of
inflammation and nutrition, has previously been recognized as an
important predictor of overall survival. mGPS, as an indicator of
systemic inflammatory response, is significantly associated with
metastatic disease. Some studies show a significantly lower
five-year survival rate of 35.2% for mGPS 2 compared to 91.5% for
mGPS 0. A higher mGPS is also associated with malnutrition. mGPS is
an independent predictor of the incidence of surgical site
infections (SSI). This complication may be associated with
anastomotic dehiscence, which not only prolongs postoperative
hospital stay but also increases the risk of disease recurrence.
[7].
Aim
The aim of this study is to evaluate the impact of nutritional
status on the postoperative course in patients with colorectal
cancer, as well as to assess the effectiveness of different
nutritional strategies in improving treatment outcomes.
Materials and Methods
This is a prospective observational study conducted at the
Department of Surgery of the Military Medical Academy from January
2024 to March 2025 in patients operated on for colorectal cancer.
The sample included patients who underwent a 7-day preoperative
nutritional preparation, and data were analyzed in the postoperative
recovery period during the same hospitalization. The inclusion
criteria for the study were: Patients with histopathologically
confirmed colorectal cancer. Patients undergoing surgical treatment.
Availability of data on preoperative nutritional status.
Demographic, clinical, and laboratory parameters were analyzed,
including anthropometric measures such as body weight and body mass
index (BMI); nutritional status assessed by NRS-2002 and SGA;
inflammatory status evaluated using mGPS; surgical data including
type of operation, duration of the procedure, and blood loss; the
Clavien–Dindo classification of postoperative complications; and
postoperative outcomes such as length of hospital stay and
complication rates.
The obtained data were analyzed using descriptive and inferential
statistics. The monitored parameters in patients with preoperative
nutritional preparation were compared, and the significance of
differences was assessed using Student’s t-test, χ² test, logistic
regression, and correlation analysis..
This methodological framework enables the assessment of the impact
of nutritional status on the postoperative course and the potential
benefits of preoperative nutritional preparation..
The study was approved by the Ethics Committee of the Military
Medical Academy under number 132/2025 on September 25, 2025.
Results
Table 1. – Parameters Monitored in the Study and
Their Mean, Minimum, and Maximum Values
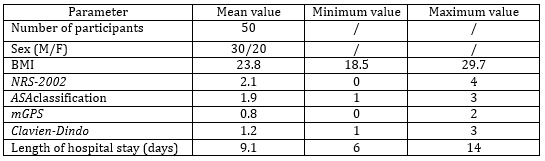
Table 2 – The height of the bars indicates the
strength of the correlation (higher |r| = stronger relationship).
Darker color indicates statistically significant associations.
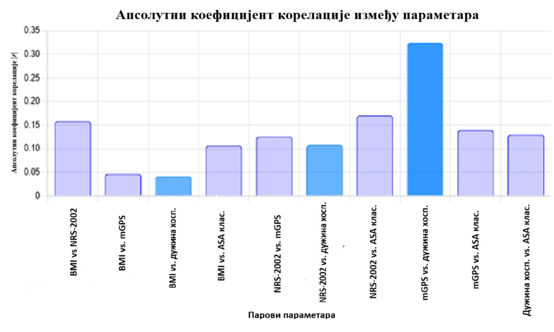
These results indicate the following relationships:
• There is a weak positive correlation between BMI and NRS-2002,
which may suggest that patients with higher BMI have a higher
nutritional risk.
• The strongest positive correlation is between mGPS and length of
hospital stay (0.3239), suggesting that inflammatory status may play
a role in the duration of hospitalization.
t-test for BMI and length of hospital stay
• t-statistic: 157.85
• p-value:
9.20 × 10⁻¹²⁰
• p-value: < 0.05
• This result indicates a statistically significant association
between BMI and length of hospital stay (p < 0.05), although the
correlation coefficient suggests a weak negative relationship.
ANOVA test for NRS-2002 and length of hospital stay
• F-statistic: 2509.59
• p-value: 1.22 × 10⁻⁷¹
• p-value: < 0.05
• There is a highly significant association between assessed
nutritional risk (NRS-2002) and postoperative length of hospital
stay, with strong statistical significance.
DISCUSSION
Colorectal cancer (CRC) is the third most common cancer and the
fourth leading cause of cancer-related death [8]. Nutritional status
is a very important indicator for predicting postoperative survival
outcomes in CRC, and monitoring nutritional status is a common
criterion used in treatment follow-up [9]. In this study, we
demonstrated that nutritional status significantly affects the
postoperative course in patients with CRC. Data analysis indicated a
trend showing that patients with higher nutritional risk (NRS-2002 ≥
3) have a longer postoperative hospital stay. Additionally, a
negative correlation was observed between BMI and length of hospital
stay, suggesting that patients with lower BMI values experience
slower postoperative recovery [10]. The results indicate that lower
BMI values, within the range of malnutrition, prolong the duration
of postoperative recovery..
Some studies suggest that a high mGPS has a negative impact on
survival in patients with colorectal cancer. In a meta-analysis,
Tsung-Hsien Wu reviewed the existing evidence on the practicality of
mGPS and confirmed its accuracy in predicting cancer prognosis.
[11].
The systemic inflammatory response in patients, measured using the
mGPS scale, showed a significant impact on the length of hospital
stay, highlighting the importance of systemic inflammation in the
postoperative period. In addition, the ASA classification was also
correlated with the length of hospitalization, with patients in
higher ASA classes experiencing longer recovery [12]. Furthermore,
results from similar studies have shown that patients with a
pronounced inflammatory response (elevated CRP, hypoalbuminemia)
have an increased risk of postoperative complications and a longer
recovery period [13]. This is consistent with our findings, where
patients with higher mGPS scores had prolonged postoperative
recovery.
Nutritional interventions, such as preoperative oral nutritional
supplementation, have shown potential in reducing postoperative
hospital stay and decreasing complication rates, which is consistent
with previous research and ESPEN guideline recommendations. [2].
Comparing the results of our study with the available literature, it
is evident that our findings are consistent with studies emphasizing
the importance of nutritional screening and interventions. A study
conducted in the United Kingdom showed that patients with
malnutrition have a 30% longer hospital stay and a higher rate of
postoperative complications compared to patients with adequate
nutritional status [14]. Similarly, a study from Germany indicated
that the implementation of an individualized nutritional protocol
can reduce postoperative infections by 25% [15]. These findings
support the importance of early nutritional intervention, which is
also confirmed by our data.
CONCLUSION
It is stated here that the nutritional status of patients with
colorectal cancer plays a key role in the postoperative course and
treatment outcomes. Early screening of nutritional status and the
implementation of individualized nutritional strategies may
contribute to reducing postoperative complications and promoting
faster patient recovery..
Further research aimed at determining the optimal duration of
preoperative nutritional therapy and the implementation of
personalized nutritional interventions is necessary in order to
further improve treatment outcomes and the quality of life of
patients with colorectal canc.
LITERATURE:
1. David GA Williams, Jeroen Molinger, Paul E Wischmeyer, The
Malnourished Surgery Patient: A Silent Epidemic in Perioperative
Outcomes?, Curr Opin Anaesthesiol. 2019 Jun;32(3):405–411.
2. Weimann A, Braga M, Carli F, Higashiguchi T, Hübner M, Klek S. et
al. ESPEN practical guideline: Clinical nutrition in surgery. Clin
Nutr. 2021 Jul;40(7):4745-4761.
3. T. Cederholm et al, ESPEN guidelines on definitions and
terminology of clinical nutrition, Clinical Nutrition, Volume 36,
Issue 1, February 2017, p49-64,
4. Deftereos I, Kiss N, Isenring E, Carter VM, Yeung JM. A
systematic review of the effect of preoperative nutrition support
onnutritional status and treatment outcomes in upper
gastrointestinal cancer resection. Eur J Surg Oncol
2020;46(8):1423-34.
5. Gupta A, Gupta E, Hilsden R, Hawel JD, Elnahas AI, Schlachta CM,
Alkhamesi NA. Preoperative malnutrition in patients with colorectal
cancer. Can J Surg. 2021 Nov 25;64(6):E621-E629.
6. Rossi S, Basso M, Strippoli A, Schinzari G, D’Argento E, Larocca
M, et al, Are markers of systemic inflammation good prognostic
indicators in colorectal cancer?, Clinical Colorectal Cancer (2017),
4-5.
7. Masano Sagawa et al. Worse Preoperative Status Based on
Inflammation and Host Immunity Is a Risk Factor for Surgical Site
Infections in Colorectal Cancer Surgery, Journal of Nippon Medical
School Vol.84 No.5
8. Inés Mármol, Cristina Sánchez-de-Diego, Alberto Pradilla Dieste,
Elena Cerrada, María Jesús Rodriguez Yoldi,Colorectal Carcinoma: A
General Overview and Future Perspectives in Colorectal Cancer, Int.
J. Mol. Sci. 2017, 18(1), 197;
9. Zhi Wang et al, Study on the correlation between controlling
nutritional status score and clinical biochemical indicators in
patients with colorectal cancer, Helyon, Volume 10, Issue 5e27202,
March 2024
10. Schwegler I, von Holzen A, Gutzwiller JP, Schlumpf R, Mühlebach
S, Stanga Z. Nutritional risk is a clinical predictor of
postoperative mortality and morbidity in surgery for colorectal
cancer. Br J Surg. 2010 Jan;97(1):92-7.
11. Tsung-Hsien Wu, Yao-Te Tsai, Kuan-Yin Chen, Wing-Keen Yap, Chih-Wei
Luan, Utility of High-Sensitivity Modified Glasgow Prognostic Score
in Cancer Prognosis: A Systemic Review and Meta-Analysis, Int. J.
Mol. Sci. 2023, 24(2), 1318;
12. Fukatsu K. Role of nutrition in gastroenterological surgery. Ann
Gastroenterol Surg. 2019 Feb 25;3(2):160-168.
13. Deutz NE, Bauer JM, Barazzoni R, Biolo G, Boirie Y,
Bosy-Westphal A, et al Protein intake and exercise for optimal
muscle function with aging: recommendations from the ESPEN Expert
Group. Clin Nutr. 2014 Dec;33(6):929-36.
14. Kondrup J, Rasmussen HH, Hamberg O, Stanga Z; Ad Hoc ESPEN
Working Group. Nutritional risk screening (NRS 2002): a new method
based on an analysis of controlled clinical trials. Clin Nutr. 2003
Jun;22(3):321-36.
15. Braga M, Gianotti L, Vignali A, Di Carlo V. Immunonutrition in
gastric cancer surgical patients. Nutrition. 1998
Nov-Dec;14(11-12):831-5.
The authors declare that they have no conflicts of interest.
|
|
|
|


