| |
|
|
INTRODUCTION Dyslipidemias encompass disorders of lipid
metabolism that significantly contribute to the development of
atherosclerotic cardiovascular disease (ASCVD). Elevated low-density
lipoprotein cholesterol (LDL-C) represents the main causal factor of
atherogenesis. Dyslipidemias are a heterogeneous group of lipid
metabolism disorders characterized by increased or decreased
concentrations of plasma lipoproteins. They are a key modifiable
risk factor for ASCVD, including coronary heart disease,
cerebrovascular disease, and peripheral vascular disease.
Epidemiological data demonstrate a clear linear relationship between
LDL cholesterol (LDL-C) levels and the incidence of cardiovascular
events, confirming LDL-C as the primary causal factor in
atherogenesis [1–7].
In apparently healthy individuals, the risk of developing ASCVD is
most often the result of the interaction of multiple risk factors.
This forms the basis for assessment and management of overall
cardiovascular (CV) risk. Risk factor screening should include lipid
profiling in men over 40 years of age and in women over 50 years of
age or after menopause. The updated electronic risk assessment
system HeartScore (www.heartscore.org), including SCORE2 and
SCORE2-OP, supports clinical decision-making in order to avoid
under- or overtreatment with lipid-lowering therapy. Certain
individuals presenting with high, very high, or extreme
cardiovascular risk do not require formal risk scoring but instead
require immediate management of all risk factors. This applies to
patients with established ASCVD, diabetes mellitus (DM), or chronic
kidney disease (CKD) stage G3b–G4 or overt chronic renal failure. It
should be noted that all risk scoring systems are relatively rigid
and require additional clinical judgment when making final
therapeutic decisions. Additional risk modifiers are included in
electronic systems such as HeartScore (www.heartscore.org). This
comprehensive approach allows flexibility, as failure to achieve
optimal risk reduction through one factor can be compensated by more
intensive control of other risk factors.
According to current global, American, and European guidelines (ADA,
AHA/ACC, ESC/EAS 2023–2026), early diagnosis and aggressive lipid
control remain central components of ASCVD prevention [3–5].
In the period 2023–2026, new versions of major international
dyslipidemia guidelines were published, including those of the
European Society of Cardiology and European Atherosclerosis Society
(ESC/EAS), the American Heart Association (AHA), the American
College of Cardiology (ACC), and the American Diabetes Association
(ADA).
All these guidelines emphasize the need for earlier, more intensive,
and combination therapy in order to achieve lower levels of
atherogenic lipoproteins, with special focus on LDL-C, non-HDL-C,
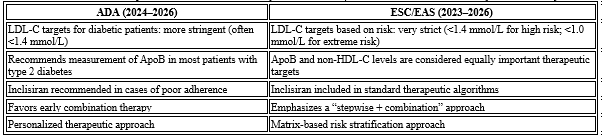
and apolipoprotein B (ApoB) [3–5] (Table 1).
Table 1. Comparative overview of key ADA vs ESC/EAS
recommendations (2023–2026)
AIM OF THE STUDY
The aim of this review article is to comprehensively present
contemporary principles of diagnosis, classification, early
screening, assessment of 10-year cardiovascular risk using SCORE2
and SCORE2-OP tools, and therapeutic management of dyslipidemias in
accordance with the latest international standards and guidelines
(ESC/EAS, ADA, AHA, ACC).
CLASSIFICATION OF DYSLIPIDEMIAS
- Primary dyslipidemias
- Familial hypercholesterolemia (FH)
- Familial combined hyperlipidemia
- Polygenic hyperlipidemia
- Secondary dyslipidemias
Most commonly caused by:
- Diabetes mellitus
- Obesity and metabolic syndrome
- Chronic kidney disease
- Hypothyroidism
- Liver diseases
- Medications (corticosteroids, antipsychotics, retinoids,
immunosuppressants) [15–17]
DIAGNOSIS OF DYSLIPIDEMIAS
Standard diagnostic approach
Diagnosis includes measurement of the standard lipid profile: total
cholesterol, LDL-C, HDL-C, and triglycerides; calculation of non-HDL-C
and ApoB; assessment of secondary causes; evaluation of global
cardiovascular risk; and review of family history.
Dyslipidemia screening is always indicated in patients with clinical
manifestations of cardiovascular disease (CVD), in clinical
conditions associated with increased cardiovascular risk, and
whenever risk factor screening is warranted. In several clinical
conditions, dyslipidemia may contribute to an increased risk of
developing CVD. Chronic autoimmune inflammatory diseases such as
rheumatoid arthritis, systemic lupus erythematosus (SLE), and
psoriasis are considered conditions associated with increased
cardiovascular risk. In addition, in women, gestational diabetes and
hypertension during pregnancy are important risk indicators, while
in men, erectile dysfunction is considered a risk marker. Patients
with chronic kidney disease and overt renal failure also have
increased cardiovascular risk, and dyslipidemia screening is
indicated in these individuals.
It is always necessary to identify clinical manifestations of
genetic dyslipidemias, such as xanthomas, xanthelasmas, and early
corneal arcus (before the age of 45), as these are indicators of
severe lipoprotein disorders, primarily familial
hypercholesterolemia (FH), which is most often a monogenic disorder
associated with premature ASCVD. Screening for dyslipidemia is also
indicated in patients with peripheral arterial disease (PAD) or in
the presence of increased carotid intima-media thickness (IMT) or
carotid plaques.
Screening should also be considered in all adult men aged ≥40 years
or women aged ≥50 years or in early postmenopause, especially in the
presence of additional risk factors. Screening is also indicated in
the offspring of patients with severe dyslipidemia, with follow-up
in specialized clinics if necessary. Furthermore, screening of
family members of patients with premature ASCVD is recommended [2].
Evaluation of Lipid and Apolipoprotein Laboratory Parameters [2]
The proposed lipid analyses used for assessment include total
cholesterol (TC), triglycerides (TG), HDL-C, and LDL-C. Blood
samples collected in the fasting state and those collected after
meals provide similar results for total cholesterol (TC), LDL
cholesterol, and HDL cholesterol. Triglycerides (TGs), however, are
influenced by food intake.
There is significant intra-individual variability in serum lipid
levels. Variations of 5–10% for TC and >20% for TG, particularly in
patients with hypertriglyceridemia (HTG), are not uncommon. This is
partly due to analytical variation, but also to external factors
such as diet, physical activity level, and seasonal variation,
including higher TC and HDL cholesterol levels during winter.
LDL cholesterol
In most clinical studies, LDL cholesterol is calculated using the
Friedewald formula [2]:
LDL-C = TC − HDL-C − TG / 2.2 (mmol/L)
LDL-C = TC − HDL-C − TG / 5 (mg/dL)
Methodological errors may accumulate because this calculation is
based on three parameters: total cholesterol (TC), triglycerides
(TG), and HDL cholesterol. Direct methods for LDL-C measurement are
available and are now widely used. In general, direct and calculated
LDL-C values show good agreement.
New LDL-C estimation formulas, such as the Martin/Hopkins and
Sampson equations, are particularly recommended in ADA and AHA/ACC
guidelines [3–4]. Direct methods for measuring HDL-C and LDL-C are
widely used and are reliable in patients with normal lipid profiles.
However, in hypertriglyceridemia (HTG), they may be unreliable, and
results can vary between commercial assays.
Lipoprotein(a) [Lp(a)]
Lipoprotein(a) [Lp(a)] has been identified in several studies as an
independent risk factor in the pathophysiology of atherosclerotic
cardiovascular disease and aortic stenosis. Lp(a) shares
similarities with LDL but contains a unique protein,
apolipoprotein(a) [apo(a)], which is structurally homologous to
plasminogen.
Lp(a) measurements are relatively stable over time. Statins do not
reduce Lp(a) levels; however, a reduction of approximately 30% has
been observed with PCSK9 (proprotein convertase subtilisin/kexin
type 9) inhibitors and nicotinic acid. However, a clear reduction in
cardiovascular outcomes through direct Lp(a) targeting has not yet
been conclusively demonstrated. Emerging therapies targeting the
Lp(a) gene can reduce circulating Lp(a) levels by more than 80%.
Lipid parameters in cardiovascular risk estimation
Most cardiovascular risk assessment systems use TC and LDL-C, while
other markers such as apoB and non-HDL-C, although physiologically
logical, are mainly supported by post-hoc analyses. TC and LDL-C
remain primary therapeutic targets, while non-HDL-C and apoB are
considered secondary targets. In patients with elevated
triglycerides, additional risk is contributed by triglyceride-rich
lipoproteins, which must be taken into account.
Total cholesterol (TC) is recommended for cardiovascular risk
estimation using the SCORE system. However, in individual cases, TC
may be misleading. This is particularly relevant in women, who often
have elevated HDL-C levels, and in patients with diabetes or
elevated triglycerides, who frequently have reduced HDL-C levels.
Overall risk assessment is not required in individuals with familial
hyperlipidemia (including FH) or in those with TC >7.5 mmol/L (290
mg/dL), as these patients are always considered high risk and
require special clinical attention.
Non-HDL cholesterol
Non-HDL cholesterol is used to estimate the total amount of
atherogenic lipoproteins in plasma, including VLDL, VLDL remnants,
intermediate-density lipoproteins (IDL), LDL, and Lp(a), and it
shows a strong correlation with ApoB levels. It is easily calculated
as:
non-HDL-C = TC − HDL-C
According to the updated ESC dyslipidemia guidelines [3], SCORE2 and
SCORE2-OP recommend non-HDL-C as a better risk indicator than LDL-C.
In several analyses, non-HDL-C has shown superiority over other
measures, while in others it provides similar information to LDL-C.
Compared with LDL-C, non-HDL-C has the advantage of simplicity and
does not require additional testing. It also includes
triglyceride-rich atherogenic lipoproteins (VLDL, IDL, and
remnants), which are increasingly recognized as important in
atherogenesis based on genetic (GWAS) evidence.
LDL-C remains the primary treatment target; however, non-HDL-C is
recommended as a secondary target once LDL-C goals are achieved. The
non-HDL-C target can be estimated by adding 0.8 mmol/L (30 mg/dL) to
the LDL-C target value.
High-density lipoprotein cholesterol (HDL-C)
Low HDL-C is an important independent cardiovascular risk factor and
is included in most risk scoring systems, including HeartScore. Very
high HDL-C levels are not necessarily protective. Epidemiological
studies define increased risk thresholds as:
Men: HDL-C < 1.0 mmol/L (40 mg/dL)
Women: HDL-C < 1.2 mmol/L (48 mg/dL)
The protective role of HDL-C has been questioned in several
Mendelian randomization studies. Recent evidence suggests that
dysfunctional HDL particles may be more relevant to atherosclerosis
development than absolute HDL-C levels.
Triglycerides (TG)
Triglycerides are measured using enzymatic methods. Rare analytical
errors may occur in patients with extreme hypertriglyceridemia,
particularly at very high TG levels. Elevated TG levels are often
associated with low HDL-C and increased numbers of small dense LDL
particles.
Multiple meta-analyses suggest that TG may represent an independent
cardiovascular risk factor. Genetic studies further support the role
of triglycerides in directly contributing to cardiovascular disease.
Recent data also suggest that non-fasting TG levels may provide
important information regarding remnant lipoproteins associated with
increased cardiovascular risk.
APOLIPOPROTEINS
There are reliable immunochemical methods for the determination
of apolipoproteins using conventional autoanalyzers. Analytical
performance is generally good, and these assays do not require
fasting conditions and are not affected by elevated triglyceride
(TG) levels.
Apolipoprotein B (ApoB)
Apolipoprotein B (ApoB) is the main apolipoprotein of the
atherogenic lipoprotein family (VLDL, IDL, and LDL). ApoB is useful
for estimating the total number of these particles in plasma. This
feature is particularly important in cases of elevated low-density
lipoprotein (LDL) concentrations. Several prospective studies have
shown that ApoB performs similarly to LDL cholesterol and non-HDL
cholesterol in predicting cardiovascular risk. Although ApoB has not
been established as a primary treatment target in clinical trials,
several post-hoc analyses suggest that it may be used not only as a
risk marker but also as a potential therapeutic target.
Apolipoprotein A1 (ApoA1)
Apolipoprotein A1 (ApoA1) is the main protein component of HDL
cholesterol and provides a reliable estimate of HDL particle
concentration. However, each HDL particle may carry between one and
five ApoA1 molecules.
Apolipoprotein CIII (ApoCIII)
Apolipoprotein CIII (ApoCIII) is recognized as a potentially
important emerging cardiovascular risk factor. ApoCIII is a key
regulator of triglyceride metabolism, and elevated serum ApoCIII
levels are associated with increased concentrations of VLDL and
serum triglycerides. In addition, loss-of-function mutations in
ApoCIII are associated with low triglyceride levels and reduced
cardiovascular risk.
GENETIC DIAGNOSTICS [4]
Genetic testing is recommended in cases of suspected familial
hypercholesterolemia (FH), extremely elevated LDL-C levels (>4.9
mmol/L or >190 mg/dL), and a family history of premature ischemic
heart disease.
THERAPEUTIC APPROACHES
The treatment of dyslipidemias is based on a combination of
non-pharmacological and pharmacological strategies, primarily aimed
at reducing LDL cholesterol, but also at controlling triglycerides,
increasing HDL cholesterol, and reducing overall atherogenic burden.
Contemporary guidelines are consistent with the key principle: “the
lower, the better” for LDL-C, especially in patients at high and
very high cardiovascular risk [1–9].
1. Non-pharmacological approaches
Lifestyle modification
These interventions represent the foundation of therapy in all
patients with dyslipidemia, regardless of risk level. The most
important measures include:
Reduction of saturated fat and trans fat intake
Mediterranean or DASH dietary pattern
Increased intake of dietary fiber and plant sterols
Aerobic physical activity ≥150 minutes per week
Weight reduction of ≥5–7% in overweight and obese patients
Smoking cessation
Reduction of alcohol intake in hypertriglyceridemia
Although lifestyle changes can reduce LDL-C by approximately 5–15%,
they are usually insufficient as monotherapy in patients at high
cardiovascular risk [16–22].
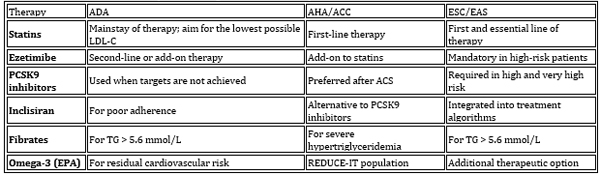
2. Pharmacological treatment of dyslipidemia (Table 2 and
Figure 1)
Table 2. Therapeutic Drug Classes — Comparison of
Guideline Recommendations
Scheme 1. Graphical representation of the
mechanism of action of lipid-lowering drugs
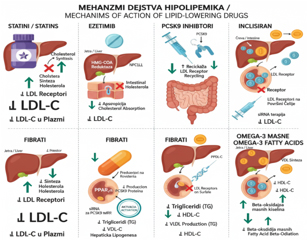
Statins (HMG-CoA reductase inhibitors)
Statins remain the first-line therapy in most patients. They are
classified into high-intensity statins (atorvastatin 40–80 mg,
rosuvastatin 20–40 mg) and moderate-intensity statins (simvastatin,
pravastatin, lovastatin, pitavastatin). Effects include LDL-C
reduction of 30–60% depending on dose and a reduction in the risk of
myocardial infarction, stroke, and cardiovascular (CV) mortality by
25–40% [8–10].n
Adverse effects include myopathy, elevated liver enzymes, and very
rarely rhabdomyolysis.
Ezetimibe (cholesterol absorption inhibitor)
Ezetimibe inhibits the NPC1L1 transporter in the small intestine,
thereby reducing cholesterol absorption.
Its clinical importance lies in its recommendation as second-line
therapy in patients whose LDL-C remains above target despite maximal
statin therapy. It provides an additional LDL-C reduction of 20–25%
and is safe and well tolerated [13].
Proprotein Convertase Subtilisin/Kexin Type 9 (PCSK9)
inhibitors
PCSK9 inhibitors are monoclonal antibodies, including evolocumab and
alirocumab. They block the PCSK9 protein, thereby increasing LDL
receptor recycling and reducing LDL-C levels by 50–65%.
Indications include:
Patients at very high risk (previous myocardial infarction,
polyvascular disease)
Patients with familial hypercholesterolemia (FH)
Patients who do not achieve target LDL-C levels despite maximal
statin + ezetimibe therapy [10–11]
Major clinical trials (FOURIER, ODYSSEY Outcomes) demonstrated a
significant reduction in cardiovascular mortality and myocardial
infarction.
Inclisiran (siRNA therapy)
Inclisiran is a small interfering RNA (siRNA) that inhibits hepatic
synthesis of PCSK9 in hepatocytes.
Advantages include administration only twice yearly and sustained
LDL-C reduction of approximately 50%, making it particularly
suitable for patients with poor adherence.
Guideline integration: ESC/EAS 2023–2026 includes inclisiran in
standard treatment algorithms for very high-risk patients, while ADA
recommends it in cases of poor therapeutic adherence [5–7].
Fibrates
Fenofibrate and bezafibrate are used in specific lipid disorders.
Indications include:
Triglycerides >5.6 mmol/L (>500 mg/dL)
Prevention of pancreatitis
Residual hypertriglyceridemia in type 2 diabetes mellitus (DM2)
Omega-3 fatty acids (EPA formulations)
High-dose EPA (2–4 g/day) is used to reduce triglyceride levels and
stabilize atherosclerotic plaque.
The REDUCE-IT trial demonstrated a reduction in cardiovascular
outcomes in patients with elevated triglycerides [39].
NEW DEVELOPMENTS IN THE 2023–2026 GUIDELINES (ADA, ESC/EAS,
AHA/ACC)
In the past three years, several important changes have occurred
that significantly impact everyday clinical practice. (Table 3 and
Scheme 2).
Scheme 2. Graphical representation of the
therapeutic algorithm and comparison of guidelines
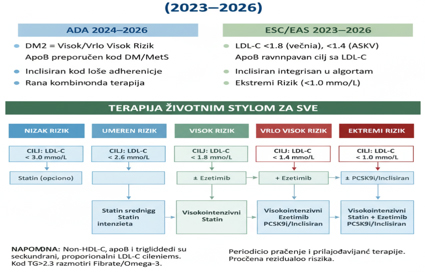
Table 3. Comparative LDL-C targets according to
guidelines (ADA, AHA/ACC, ESC/EAS)

1. ADA 2024–2026 – Diabetes and dyslipidemia
Patients with type 2 diabetes mellitus (DM2) are automatically
classified as having high or very high cardiovascular risk. The LDL-C
target in most diabetic patients is <1.8 mmol/L, while in patients
with ASCVD the target is <1.4 mmol/L. Measurement of apoB is
recommended in individuals with obesity, metabolic syndrome, and
high triglycerides. Inclisiran is recommended in cases of poor
adherence. The emphasis is on early initiation of combination
therapy.
2. AHA/ACC 2023–2025 – Personalized therapeutic approach
PCSK9 inhibitors are increasingly used even after the first
myocardial infarction. The LDL-C goal is “the lower, the better,”
although in some situations there is no strict numerical target.
Lifelong LDL-C monitoring is emphasized in patients with familial
hypercholesterolemia (FH). Non–HDL-C has an important role in
patients with elevated triglycerides.
3. ESC/EAS 2023–2026 – Most aggressive LDL-C approach
A new “extreme risk” category has been introduced (e.g.,
polyvascular disease, recurrent ACS). LDL-C targets are: high risk
<1.8 mmol/L, very high risk <1.4 mmol/L, extreme risk <1.0 mmol/L.
LDL-C, non–HDL-C, and apoB are considered equal target parameters.
Inclisiran is formally included in the therapeutic algorithm. There
is a stronger focus on reduction of remnant lipoproteins and
triglycerides.
The comparative therapeutic algorithm according to cardiovascular
risk level is shown in Tables 4 and 5.
Table 4. Comparative therapeutic algorithm
according to cardiovascular risk level
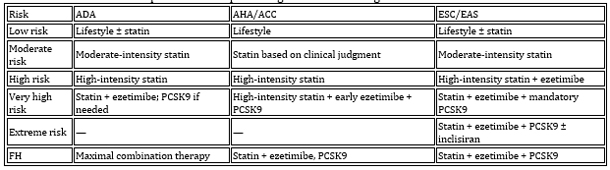
Table 5. Recommendations for specific populations
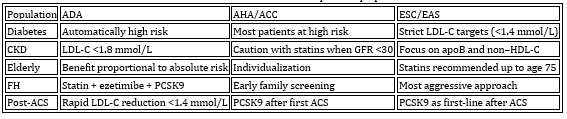
1. Effect of LDL cholesterol reduction on cardiovascular
outcomes
In a large meta-analysis by the Cholesterol Treatment Trialists'
Collaboration, including over 170,000 patients, it was shown that
each reduction of LDL-C by 1 mmol/L (~39 mg/dL) reduces the risk of
major vascular events by approximately 22% [23]. This effect is
consistent across men and women, younger and older individuals,
patients with diabetes, those with prior myocardial infarction, and
in both primary and secondary prevention. LDL-C reduction is
beneficial in almost all clinical populations.
2. Statins – evidence from clinical trials
High-intensity statins have been shown to reduce myocardial
infarction by 25–35%, ischemic stroke by 20–30%, and cardiovascular
mortality by 15–20% [6–8]. In addition to LDL reduction, statins
exert pleiotropic effects, including plaque stabilization,
anti-inflammatory action, and improvement of endothelial function
[24].
3. Ezetimibe – clinical outcomes
The IMPROVE-IT trial (ezetimibe + statin) demonstrated an additional
LDL-C reduction of ~23% and a 6% relative reduction in primary
cardiovascular outcomes (p=0.016) [11]. This supports the importance
of combination therapy.
4. PCSK9 inhibitors – greatest benefit in highest-risk
patients
The FOURIER trial (evolocumab) showed LDL-C reduction to ~0.8 mmol/L,
a 15% reduction in cardiovascular events, and a 27% reduction in
myocardial infarction risk [10]. The ODYSSEY Outcomes trial (alirocumab)
demonstrated a significant reduction in cardiovascular mortality
after acute coronary syndrome [11]. The key principle is: the lower
the LDL-C, the greater the benefit.
5. Inclisiran – a new era of long-term lipid control
Inclisiran, an siRNA-based therapy, provides sustained and stable
LDL-C reduction. Its advantages include dosing every 6 months,
improved adherence, and approximately 50% LDL-C reduction [12].
Large outcome trials are still ongoing, but current data are
promising.
6. Triglycerides and residual risk
Elevated triglycerides and remnant lipoprotein particles
significantly increase cardiovascular risk, particularly in patients
with diabetes and metabolic syndrome [19–20]. The REDUCE-IT trial (EPA
4 g/day) demonstrated a 25% reduction in major cardiovascular events
and a 20% reduction in cardiovascular mortality [39]. This confirms
that residual risk is important and that LDL-C reduction alone is
not sufficient.
LATEST AMERICAN RECOMMENDATIONS FOR THE MANAGEMENT OF
DYSLIPIDEMIA (2026)
The new American recommendations [40] reintroduce a focus on LDL
cholesterol target values: for patients with borderline or
intermediate risk, the target is <2.6 mmol/L; for high-risk patients
<1.8 mmol/L; and for very high-risk patients (i.e., in secondary
prevention), LDL-C should be reduced to <1.4 mmol/L.
The PREVENT-ASCVD calculator has been adopted, which estimates both
10-year and 30-year risk of adverse cardiovascular events
(myocardial infarction, stroke, or cardiovascular mortality). Key
patient characteristics used for risk estimation include age, sex,
systolic blood pressure, antihypertensive therapy, presence of
diabetes, smoking status, and laboratory markers (total and LDL
cholesterol). These variables are required for the basic model. The
expanded model additionally includes body mass index (BMI) and
kidney function, while the full model also incorporates albuminuria
and HbA1c [40].
A notable innovation is the assessment of both 10-year and 30-year
cardiovascular risk in individuals aged 30 to 59 years. The new
recommendations also strengthen the use of additional biomarkers for
assessing residual cardiovascular risk, such as lipoprotein(a),
which is recommended to be measured at least once in a lifetime, and
apolipoprotein B, which is particularly useful in patients with
diabetes and hypertriglyceridemia.
Greater emphasis is placed on the assessment of coronary artery
calcium (CAC) score [40], especially in situations where the
decision to initiate statin therapy is uncertain—most commonly in
patients with borderline or intermediate risk.
An important aspect of the new recommendations is earlier and more
intensive LDL-C reduction. This is based on the concept that
cumulative exposure to elevated LDL-C determines cardiovascular
risk. Therefore, earlier initiation of therapy reduces lifetime
exposure to high LDL-C levels and leads to a greater reduction in
cardiovascular risk.
Accordingly, a complementary recommendation is universal lipid
screening and lifelong prevention. Rather than waiting for
cardiovascular events to occur, periodic assessment of lipid status
is advised, with particular attention to screening in patients with
diabetes, cardio-renal-metabolic syndrome, and in children [40].
PROGNOSIS
Dyslipidemias, if not properly diagnosed and treated,
significantly increase the risk of atherosclerotic cardiovascular
disease, premature disability, and mortality. However, contemporary
therapeutic approaches allow for a substantial improvement in
prognosis.
1. Prognosis
Prognosis depends on baseline LDL-C levels, the presence of
comorbidities, degree of adherence, genetic factors (especially in
familial hypercholesterolemia), and the timeliness of therapy
initiation. In patients who achieve LDL-C target levels according to
ESC/EAS guidelines (<1.4 mmol/L for high-risk patients), the risk of
new cardiovascular events can be reduced by up to 50% [5].
2. Clinical implications
Recent guidelines (2023–2026) emphasize the following principles:
LDL-C is the primary therapeutic target; lower LDL-C equals better
prognosis. Combination therapy is the rule rather than the exception
(statin + ezetimibe + PCSK9 inhibitor / inclisiran). ApoB and non–HDL-C
are equally important as LDL-C, particularly in patients with
elevated triglycerides and diabetes. Personalization of therapy is
essential, with different targets for different risk categories.
CARDIOVASCULAR BENEFITS AND OUTCOMES
Reduction of atherogenic lipoproteins, particularly LDL
cholesterol, represents the most effective pharmacological strategy
in the prevention of atherosclerotic cardiovascular disease (ASCVD).
Numerous randomized clinical trials, genetic analyses, and
meta-analyses demonstrate a clear causal relationship between LDL-C
reduction and decreased risk of major cardiovascular events
[8–12,25–34].
1. Effect of LDL cholesterol reduction on cardiovascular
outcomes
In a large meta-analysis by the Cholesterol Treatment Trialists'
Collaboration, including over 170,000 patients, each 1 mmol/L (~39
mg/dL) reduction in LDL-C was associated with approximately a 22%
reduction in major vascular events [25]. This effect is consistent
across men and women, younger and older individuals, patients with
diabetes, those with prior myocardial infarction, and in both
primary and secondary prevention. LDL-C lowering is beneficial in
nearly all clinical populations.
2. Statins – evidence from clinical trials
High-intensity statins have been shown to reduce myocardial
infarction by 25–35%, ischemic stroke by 20–30%, and cardiovascular
mortality by 15–20% [8–10]. In addition to LDL-C reduction, statins
exert pleiotropic effects, including plaque stabilization,
anti-inflammatory action, and improvement of endothelial function
[26].
3. Ezetimibe – clinical outcomes
The IMPROVE-IT trial (ezetimibe + statin) demonstrated an additional
LDL-C reduction of ~23% and a 6% relative reduction in primary
cardiovascular outcomes (p=0.016) [11], supporting the importance of
combination therapy.
4. PCSK9 inhibitors – greatest benefit in highest-risk
patients
The FOURIER trial (evolocumab) showed LDL-C reduction to ~0.8 mmol/L,
a 15% reduction in cardiovascular events, and a 27% reduction in
myocardial infarction risk [8]. The ODYSSEY Outcomes trial (alirocumab)
demonstrated a significant reduction in cardiovascular mortality
after acute coronary syndrome [9]. The key principle is: the lower
the LDL-C, the greater the benefit.
5. Inclisiran – a new era of long-term lipid control
Inclisiran, as an siRNA-based therapy, provides sustained and stable
LDL-C reduction. Its advantages include administration every 6
months, improved adherence, and approximately 50% LDL-C reduction
[12]. Large outcome trials are still awaited, but current data are
promising.
6. Triglycerides and residual risk
Elevated triglycerides and remnant lipoprotein particles
significantly increase cardiovascular risk, particularly in patients
with diabetes and metabolic syndrome [19–20]. The REDUCE-IT trial (EPA
4 g/day) demonstrated a 25% reduction in major cardiovascular events
and a 20% reduction in cardiovascular mortality [39]. This confirms
that residual risk is important and that LDL-C reduction alone is
not sufficient.
GENE THERAPY AND FUTURE PERSPECTIVES [41]
In vivo gene or base editing represents a novel therapeutic
strategy currently being investigated for the treatment of
dyslipidemia, targeting genes such as PCSK9 and ANGPTL3. VERVE-101
is an experimental CRISPR-based therapy that includes mRNA encoding
an adenine base editor targeting the PCSK9 gene, with the aim of
permanently “silencing” (inactivating) this gene [42].
In non-human primates, a single infusion of VERVE-101 resulted in a
69% reduction in LDL-C, with sustained effects lasting up to 476
days post-dose, without significant adverse events. Following
confirmed efficacy in primates, the first human study included 10
patients with heterozygous familial hypercholesterolemia (HeFH) and
a mean LDL-C level of 201 mg/dL. A single intravenous infusion of
CRISPR-based gene therapy delivered via targeted lipid nanoparticles
resulted in up to a 55% reduction in LDL-C [43].
A next-generation therapy, Verve-102, represents an improved PCSK9
base-editing approach with enhanced liver targeting and redesigned
lipid nanoparticles, and is currently in clinical development.
Today, numerous effective strategies are available to manage
lipid-related cardiovascular risk factors. Even more promising
innovations suggest continued progress in this field. However, the
implementation of proven therapies, patient acceptance, adherence to
treatment, and ensuring equitable access to modern therapeutic
advances remain key challenges that must be addressed [4].
CONCLUSION
Dyslipidemias remain one of the most important risk factors for
atherosclerosis and cardiovascular mortality. Reduction of
atherogenic lipoproteins, particularly LDL cholesterol, represents
the most effective pharmacological strategy for the prevention of
atherosclerotic cardiovascular disease (ASCVD). Numerous randomized
clinical trials, genetic analyses, and meta-analyses demonstrate a
clear causal relationship between LDL-C reduction and a decreased
risk of major cardiovascular events.
Advances in the understanding of lipid metabolism and the
availability of novel therapies have significantly improved
treatment options. The integration of recommendations from ADA,
AHA/ACC, and ESC/EAS enables an optimal and individualized approach,
particularly in patients at high and very high risk.
A key novelty of the 2026 ACC/AHA guidelines is the assessment not
only of 10-year cardiovascular risk (fatal and nonfatal), but also
of 30-year risk in individuals aged 30 to 59 years. The new
recommendations further emphasize the use of additional biomarkers
for assessing residual cardiovascular risk, such as lipoprotein(a),
apolipoprotein B, non–HDL cholesterol, coronary artery calcium
score, body mass index, and HbA1c. Apolipoprotein B is particularly
useful in patients with diabetes and hypertriglyceridemia.
Continued research in lipidology, the development of new therapeutic
agents, and advances in genetic diagnostics will contribute to even
more effective prevention of ASCVD in the future.
REFERENCE:
1. European Society of Cardiology. ESC Guidelines for the
Management of Myocarditis and Pericarditis. Eur Heart J. 2025.
2. European Society of Cardiology. 2015 ESC Guidelines on
Pericardial Diseases. Eur Heart J. 2015;36:2921–2964.
3. American College of Cardiology. ACC Expert Consensus Decision
Pathway on Myocarditis. J Am Coll Cardiol. 2024.
4. American College of Cardiology. ACC Consensus on Pericardial
Diseases. J Am Coll Cardiol. 2024.
5. Caforio ALP, Pankuweit S, Arbustini E, et al. Current state of
knowledge on aetiology, diagnosis and therapy of myocarditis. Eur
Heart J. 2022;43:399–421.
6. Ammirati E, Frigerio M, Adler ED, et al. Management of acute
myocarditis and chronic inflammatory cardiomyopathy. Circulation.
2022;145:1722–1737.
7. Cooper LT. Myocarditis. N Engl J Med. 2009;360:1526–1538.
8. Tschöpe C, Ammirati E, Bozkurt B, et al. Myocarditis and
inflammatory cardiomyopathy: current evidence and future directions.
Nat Rev Cardiol. 2021;18:169–193.
9. Kindermann I, Barth C, Mahfoud F, et al. Update on myocarditis. J
Am Coll Cardiol. 2022;79:1326–1342.
10. Friedrich MG, Sechtem U, Schulz Menger J, et al. Cardiovascular
magnetic resonance in myocarditis: a JACC White Paper. J Am Coll
Cardiol. 2009;53:1475–1487.
11. Ferreira VM, Schulz Menger J, Holmvang G, et al. CMR mapping
techniques for myocarditis: consensus statement. JACC Cardiovasc
Imaging. 2018;11:155 169.
12. Bastać D, Marković Z, Vasić B, et al. Dijastolni stres test u
proceni bolesnika sa srčanom insuficijencijom. Zbornik radova
Zdravstvenog centra Zaječar. 2017;1:45–49.
13. Bastać D, Vasić B, Nešović P, et al. Ehokardiografska procena
dijastolne funkcije kod bolesnika sa miokarditisom. Timočki
medicinski glasnik. 2018;43(2):73–78.
14. Bastać D, Vasić B, Marković Z, et al. Srčana insuficijencija sa
očuvanom ejekcionom frakcijom – dijagnostički izazovi. Timočki
medicinski glasnik. 2020;45(1):23–28.
15. Heymans S, Cooper LT, De Smet B, et al. Inflammatory
cardiomyopathy. Eur Heart J. 2020;41:1358–1376.
16. Schultheiss HP, Kühl U, Cooper LT. Viral myocarditis. Nat Rev
Cardiol. 2021;18:145–161.
17. Imazio M, Klingel K, Kindermann I, et al. Myocarditis
management – current trends and future perspectives. Eur Heart J.
2022;43:1012–1027.
18. Kühl U, Pauschinger M, Noutsias M, et al. Viral persistence in
myocardium is associated with progressive cardiac dysfunction.
Circulation. 2005;112:1965–1970.
19. Ammirati E, Veronese G, Bottiroli M, et al. Contemporary
management of myocarditis: diagnostic and therapeutic updates. Eur J
Heart Fail. 2024;26:1–15.
20. Ammirati E, Cipriani M, Lilliu M, et al. Clinical presentation
and outcome of myocarditis. Eur J Heart Fail. 2020;22:2117–2125.
21. McCarthy RE III, Boehmer JP, Hruban RH, et al. Long term outcome
of fulminant myocarditis as compared with acute (nonfulminant)
myocarditis. N Engl J Med. 2000;342:690–695.
22. Blauwet LA, Cooper LT. Myocarditis. Prog Cardiovasc Dis.
2010;52:274–288.
23. Frustaci A, Russo MA, Chimenti C. Randomized study on the
efficacy of immunosuppressive therapy in virus negative inflammatory
cardiomyopathy. N Engl J Med. 2009;360:1526–1538.
24. Cooper LT, Berry GJ, Shabetai R. Giant cell myocarditis. N Engl
J Med. 1997;336:1860–1866.
25. Siripanthong B, Nazarian S, Muser D, et al. Recognizing COVID
19–related myocarditis: the possible pathophysiology and proposed
diagnostic algorithm. Heart Rhythm. 2020;17:1463–1471.
26. Puntmann VO, Carerj L, Wieters I, et al. Outcomes of
cardiovascular magnetic resonance imaging in patients recently
recovered from COVID 19. JAMA Cardiol. 2020;5:1265–1273.
27. Mevorach D, Anis E, Cedar N, et al. Myocarditis after
BNT162b2 mRNA COVID 19 vaccination. N Engl J Med.
2021;385:2140–2149.
28. Witberg G, Barda N, Hoss S, et al. Myocarditis after COVID 19
vaccination in patients under 40 years. N Engl J Med.
2021;385:1813–1822.
29. Bozkurt B, Kamat I, Hotez PJ. Myocarditis with COVID 19 mRNA
vaccines. Circulation. 2021;144:471–484.
30. Oster ME, Shay DK, Su JR, et al. Myocarditis cases reported
after mRNA based COVID 19 vaccination in the US. JAMA.
2022;327:331–340.
|
|
|
|







