|
||||||||||||||||||||||||||||||||||||
| [
Contents
] [ INDEX ]
|
||||||||||||||||||||||||||||||||||||
|
Review article UPDATES IN THE 2025 ESC GUIDELINES FOR MYOCARDITIS AND PERICARDITIS: AN INTEGRATIVE APPROACH TO INFLAMMATORY MYOPERICARDIAL SYNDROMES AND IMPLICATIONS FOR CLINICAL PRACTICE Dušan Bastać (1), Zoran Joksimović (1), Mila Bastać (2), Pavle Nešović (1) (1) INTERNAL MEDICINE PRACTICE "DR BASTAĆ" ZAJEČAR; (2) MEDSCAN TADIĆ DIJAGNOSTIKA, ZAJEČAR |
||||||||||||||||||||||||||||||||||||
|
|
||||||||||||||||||||||||||||||||||||
| Download in pdf format | Abstract:
Introduction: Myocarditis is an inflammatory disease of the
myocardium that can present with highly heterogeneous clinical
manifestations, ranging from asymptomatic forms to fulminant heart
failure and sudden cardiac death. The aim of this review paper is to
present contemporary diagnostic and therapeutic approaches according
to the latest ESC recommendations, integrating clinical experience
and emphasizing the need for further research. LITERATURE REVIEW:
In 2025, the European Society of Cardiology (ESC) published the
first integrated guidelines addressing the diagnosis and treatment
of myocarditis and pericarditis, introducing the new concept of
inflammatory myopericardial syndrome (IMPS). This umbrella term
recognizes the frequent clinical overlap between these two entities
and their shared pathophysiological mechanisms. The paper analyzes
key updates in classification, diagnostics, genetic evaluation, and
therapeutic approaches, with particular emphasis on revised cardiac
magnetic resonance (CMR) criteria (Lake Louise criteria), expanded
indications for endomyocardial biopsy (EMB), and innovations in the
treatment of pericarditis, including interleukin-1 inhibitors. The
diagnostic paradigm for myocarditis has been changed. The discussion
includes a comparison of ESC recommendations with American ACC/AHA
guidelines, as well as contributions from domestic literature,
particularly studies in the field of diastolic stress testing and
inflammatory cardiomyopathies. The COVID-19 pandemic has further
highlighted myocarditis as a potential complication of viral
infections. CONCLUSION: The contemporary approach to
myocarditis is shifting the paradigm by introducing the concept of
inflammatory myopericardial syndrome and involves integrated
diagnostics and therapy in accordance with the latest ESC
guidelines, recognizing this syndrome as a clinically significant
entity. Advances in the use of cardiac magnetic resonance imaging,
broader indications for endomyocardial biopsy, and the introduction
of targeted therapies, including interleukin-1 inhibitors, enable
more precise diagnosis and individualized treatment strategies.
Despite these advances, the heterogeneity of clinical presentation
remains a challenge in everyday practice. Further research is
necessary to improve understanding of the pathophysiology and to
optimize the treatment of these patients.. Key words: Myocarditis, pericarditis, inflammatory myopericardial syndrome, ESC guidelines, cardiac magnetic resonance imaging, COVID-19, endomyocardial biopsy. |
|||||||||||||||||||||||||||||||||||
|
INTRODUCTION The 2025 ESC guidelines represent a turning
point in the approach to inflammatory heart diseases, unifying
myocarditis and pericarditis within a single framework [1]. This
decision stems from an increasingly clear understanding that these
two entities are functionally, anatomically, and
pathophysiologically closely related, and that treating them
separately often leads to fragmentation in diagnosis and therapy.
The new concept of inflammatory myopericardial syndrome (IMPS)
serves as an umbrella term encompassing a clinical continuum ranging
from isolated myocarditis, through combined myopericarditis and
perimyocarditis, to isolated pericarditis, including complex mixed
forms, up to chronic inflammatory cardiomyopathy and constrictive
pericarditis [2–5, 6–8]. Such an integrative approach aims to
improve collaboration among specialists and guide future research. EPIDEMIOLOGY AND CLASSIFICATION The guidelines report an incidence of pericarditis ranging from 3
to 32 cases per 100,000 inhabitants per year, while the incidence of
myocarditis is between 6 and 8 cases per 100,000 inhabitants [1,15].
Higher rates have been observed in men and younger adults. A
particular challenge is the fact that a large number of subclinical
and mild cases, including those diagnosed within the context of
MINOCA (myocardial infarction with non-obstructive coronary
arteries), remain undiagnosed [6,9], which may lead to
underestimation of the true incidence and prevalence of chronic
forms of the disease. ETIOLOGY AND PATHOPHYSIOLOGY The etiology of myocarditis and pericarditis is heterogeneous. In
developed countries, viral infections predominate (enteroviruses,
adenoviruses, parvovirus B19, human herpesvirus 6, influenza virus,
hepatitis C virus) [5,16,17], whereas in endemic regions
tuberculosis remains an important cause of pericarditis,
particularly in predisposed individuals with HIV infection.
Bacterial causes (diphtheria, borreliosis, staphylococcal
infections) are less common. Autoimmune mechanisms may lead to
inflammation in the context of systemic diseases (lupus, sarcoidosis,
vasculitis), while toxic agents (anthracyclines, alcohol, cocaine)
and drugs (checkpoint inhibitors) can also induce myocarditis. A
genetic basis plays an important role in susceptibility to viral
infections and in determining the severity of the clinical
presentation, with variants in sarcomeric and desmosomal genes being
associated with myocarditis, and autoinflammatory diseases with
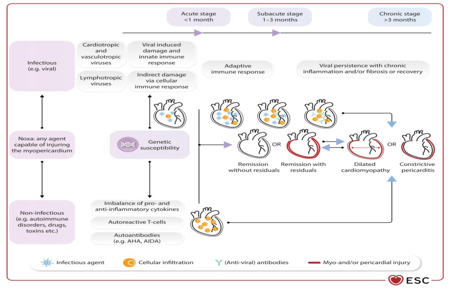
recurrent pericarditis [6,8,15]. Figure 1. Stages of inflammatory myopericardial syndrome. AHA – anti-cardiac antibodies; AIDA – antibodies against the intercalated disc. Adapted from: Eur Heart J, Volume 46, Issue 40, 21 October 2025, Pages 3952–4041, https://doi.org/10.1093/eurheartj/ehaf192
CLINICAL PRESENTATION The clinical presentation of IMPS is highly
variable. According to the time course, myocarditis is classified
into: acute (≤4 weeks), subacute (4–12 weeks), and chronic (>3
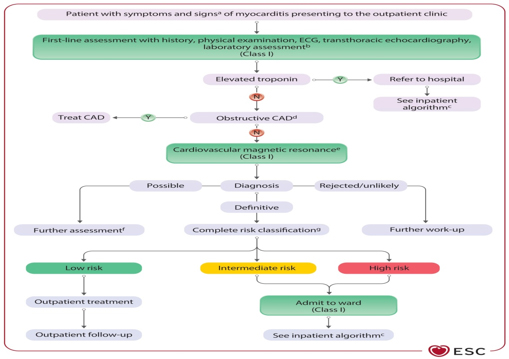
months, with persistent inflammation and remodeling). Figure 2. Diagnostic algorithm and triage for outpatients with myocarditis. Adapted from: Eur Heart J, Volume 46, Issue 40, 21 October 2025, Pages 3952–4041, https://doi.org/10.1093/eurheartj/ehaf192
The term fulminant myocarditis [21] is reserved for
patients presenting with cardiogenic shock and the most severe form
of the disease, which often requires intensive treatment and
mechanical circulatory support. DIAGNOSTIC APPROACH The 2025 ESC recommendations significantly reshape
the essence of the diagnostic pathway and disease staging,
reflecting a paradigm shift in the diagnostic process. This is
largely driven by the major role of cardiac magnetic resonance
imaging (CMR), which has become the gold standard for diagnosing
myocarditis. At the same time, the role of endomyocardial biopsy has
been refined and is now mainly reserved for severe, unclear, or
high-risk cases, as well as for guiding appropriate therapy based on
pathological and histological characterization with
immunohistochemistry and PCR detection of viral genomes in the
myocardium. Table 1. Diagnostic criteria and classification
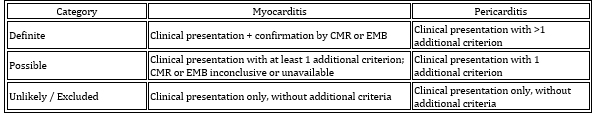
for inflammatory myopericardial syndrome (IMPS)
Table 2. Additional criteria (in addition to clinical presentation)
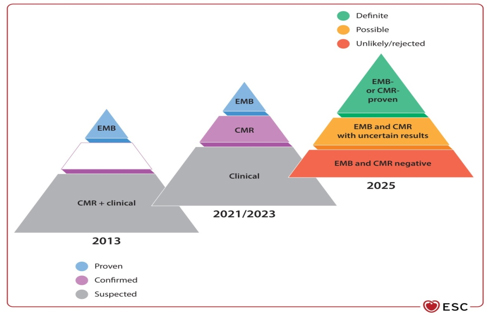
Figure 3. Paradigm shift in the clinical diagnosis of myocarditis
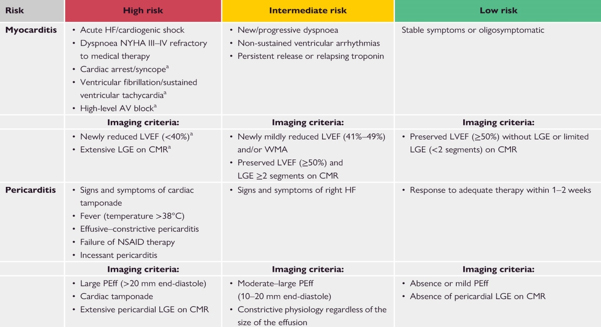
Basic diagnostic elements Table 3. Risk stratification algorithm and triage of patients with suspected myocarditis and pericarditis in outpatient settings. Hospitalization is recommended for all patients with myocarditis and high-risk pericarditis.
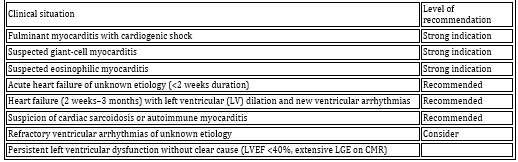
Invasive diagnostics – Endomyocardial biopsy (EMB) TABLE 4. Indications for endomyocardial biopsy (EMB) according to ESC 2025 [21]
GENETIC TESTING There is increasing evidence of an association between myocarditis and inherited cardiomyopathies. The ESC 2025 guidelines recommend genetic testing in selected patients with familial forms and recurrent pericarditis [1]. Studies highlight mutations in desmosomal genes as part of an inherited predisposition [1], particularly in patients with high diagnostic yield, such as those with:
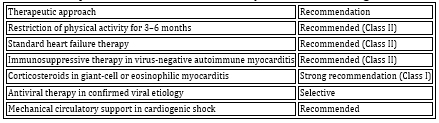
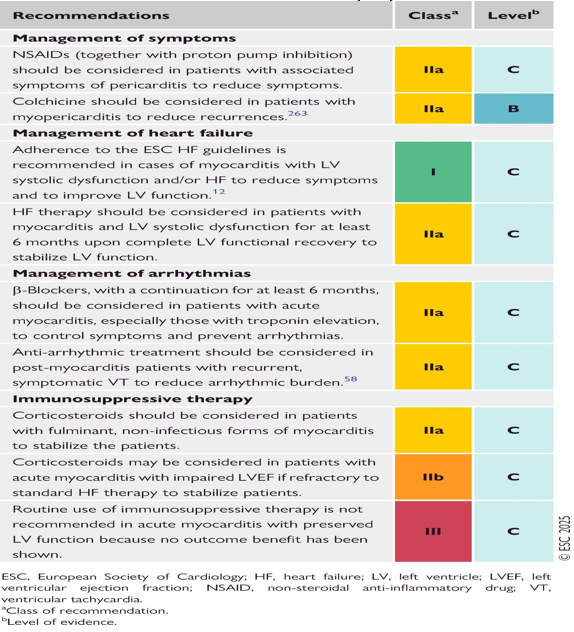
THERAPY OF MYOCARDITIS Therapeutic management is individualized and depends on etiology,
clinical presentation, and hemodynamic status [1,6] (Table 5). In
stable patients, analgesics and NSAIDs are used, with the addition
of colchicine when pericardial symptoms are present [1,31]. In heart
failure, standard HF guideline-directed medical therapy is applied
(ACE inhibitors/ARNI, beta-blockers, mineralocorticoid receptor
antagonists, SGLT2 inhibitors) [6,29].
Table 5. Therapeutic recommendations for myocarditis according to ESC 2025
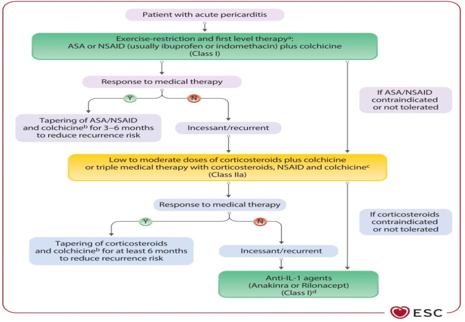
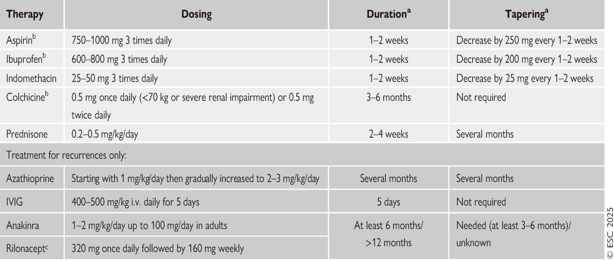
TREATMENT OF PERICARDITIS First-line therapy: Aspirin or NSAIDs in combination with
colchicine for at least 3 to 6 months (Class I A recommendation)
[1–4].
Figure 4. Proposed algorithm for pharmacological treatment of pericarditis in adults (excluding interventional procedures and pericardiectomy). Adapted from: Eur Heart J, Volume 46, Issue 40, 21 October 2025, Pages 3952–4041, https://doi.org/10.1093/eurheartj/ehaf192
TABLE 6. Therapeutic protocol for the treatment of pericarditis. Adapted from: Eur Heart J, Volume 46, Issue 40, 21 October 2025, Pages 3952–4041, https://doi.org/10.1093/eurheartj/ehaf192
TABLE 7. SUMMARY OF ESC RECOMMENDATIONS FOR THE TREATMENT OF INFLAMMATORY MYOPERICARDIAL SYNDROME (IMPS)
SPECIAL POPULATIONS Post-COVID myocarditis: May occur as a result of direct viral
infection, systemic inflammatory response, or immune dysregulation
(25–26). Diagnosis is often established by CMR, and therapy is
mainly supportive. Post-COVID myocarditis and post-vaccination forms
have been analyzed in several studies [23–28]. Rare cases of
post-vaccination myocarditis after mRNA vaccines have been reported,
most commonly in young males within several days after the second
dose. The clinical course is usually mild, and the prognosis is
favorable. The benefits of vaccination far outweigh the risks
[27–30]. PROGNOSIS AND FOLLOW-UP Prognosis depends on initial clinical presentation and etiology.
The most important predictor of adverse outcome is biventricular
dysfunction. Most patients with mild disease achieve full recovery.
In a minority of cases, progression to dilated cardiomyopathy and
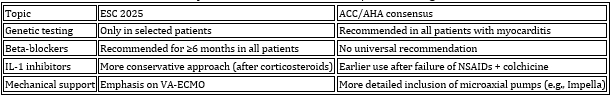
chronic heart failure may occur [14,15]. DISCUSSION The ESC 2025 guidelines represent a significant conceptual
advance with the introduction of IMPS, but also raise several
controversies. Table 8. Comparison of ESC 2025 and ACC/AHA 2024 guidelines
The ESC 2025 guidelines introduce the IMPS concept, emphasize the
role of CMR, and expand indications for EMB [1,10,17]. In the
context of local clinical practice, the studies by the author of
this literature review, Dr Dušan Bastać, significantly help bridge
gaps in the practical application of diagnostic methods for
inflammatory myopericardial syndrome in Serbia [12–14]. Figure 5. Central illustration of the ESC guidelines on myocarditis and pericarditis. Adapted from: Eur Heart J, Volume 46, Issue 40, 21 October 2025, Pages 3952–4041, https://doi.org/10.1093/eurheartj/ehaf192
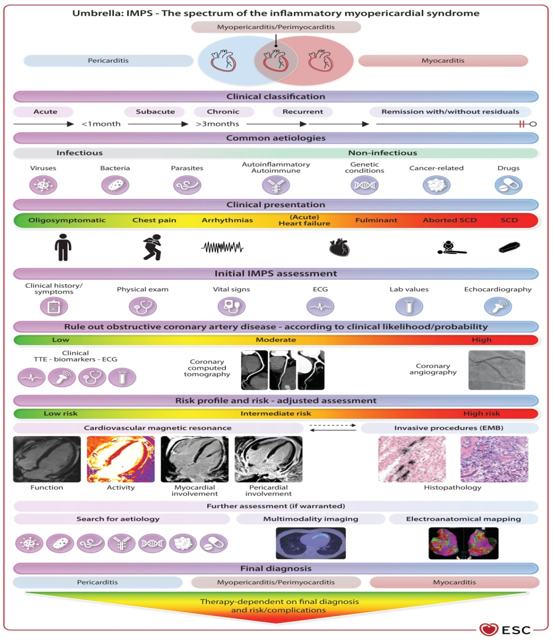
CONCLUSION The ESC 2025 recommendations and guidelines represent the most
comprehensive document to date, unifying myocarditis and
pericarditis into a single concept—inflammatory myopericardial
syndrome (IMPS), as illustrated in the figure (Figure 5: Central
illustration of ESC guidelines on myocarditis and pericarditis). REFERENCE: 1. European Society of Cardiology. ESC Guidelines for the
Management of Myocarditis and Pericarditis. Eur Heart J. 2025. |
||||||||||||||||||||||||||||||||||||
|
|
||||||||||||||||||||||||||||||||||||
| [
Contents
] [ INDEX ]
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||