| |
|
|
INTRODUCTION
Irritable Bowel Syndrome (IBS) is a chronic disorder of gut–brain
interaction, clinically characterized by recurrent abdominal pain
associated with changes in stool frequency and/or consistency.
According to the modern classification of functional
gastrointestinal disorders, IBS belongs to the group of disorders of
gut–brain interaction, in which symptoms occur in the absence of
structural, biochemical, or inflammatory abnormalities that would
fully explain them. The diagnosis is based on clinical criteria,
with exclusion of alarm features suggestive of organic disease [1].
Although traditionally labeled as a “functional” disorder,
contemporary research indicates that IBS is not synonymous with the
absence of pathophysiological changes, but rather reflects
dysregulation at multiple levels—neural, immune, microbiological,
and neuroendocrine. The absence of clear morphological findings has
often led to underestimation of the severity of the syndrome,
despite its impact on quality of life being comparable to that of
chronic inflammatory bowel diseases and other long-term conditions
[2].
IBS is a chronic condition with a fluctuating course, characterized
by periods of exacerbation and remission. Symptoms often include
bloating, a sensation of incomplete evacuation, urgency, or
straining during defecation, and their severity does not necessarily
correlate with objective laboratory or endoscopic findings. This
discrepancy between subjective symptom intensity and minimal
diagnostic abnormalities represents one of the main challenges in
everyday clinical practice.
A particular dimension of the syndrome is the high frequency of
extraintestinal manifestations. Patients commonly report chronic
pain syndromes, including fibromyalgia, chronic pelvic pain, and
tension-type headaches, as well as chronic fatigue syndrome,
temporomandibular joint disorders, non-cardiac chest pain, and
urinary symptoms. Anxiety and depressive disorders are also more
prevalent in this population. This comorbidity suggests shared
mechanisms of central sensitization and altered pain processing,
further supporting the systemic nature of the disorder..
The modern approach to IBS shifts the focus from purely
gastrointestinal symptoms toward a broader concept of dysregulation
of the brain–gut axis. The interaction between the enteric nervous
system, autonomic regulation, microbiota, immune response, and
psychological factors shapes the individual disease phenotype. This
understanding has direct therapeutic implications, as it explains
why a universal treatment approach often yields limited results and
why personalization of therapy is of particular importance [3].
Although IBS does not increase mortality nor lead to progressive
intestinal damage, its public health impact is significant. The
syndrome is associated with frequent medical consultations,
extensive diagnostic workups, and reduced work productivity. The
psychological burden, stigmatization, and chronic nature of symptoms
further impair patients’ quality of life [4].
Understanding IBS requires an integrative approach that combines
clinical criteria, epidemiological data, and contemporary insights
into pathophysiological mechanisms. In this context, irritable bowel
syndrome should not be viewed as a single disease, but rather as a
heterogeneous clinical entity with varying dominant mechanisms,
which opens the door to more precise diagnostic and therapeutic
strategies.
Epidemiology Irritable Bowel Syndrome (IBS) is one of the
most common gastrointestinal disorders in the general population.
Population-based studies estimate the prevalence of IBS at 10% to
15%, with an annual incidence of approximately 1–2%. Prevalence
varies significantly between countries. The lowest prevalence has
been reported in France (1.1%), while the highest has been reported
in Mexico (35.5%); a prevalence of 7.1% has been observed in the
USA, Europe, Australia, and New Zealand.
Approximately 50% of individuals with IBS report symptom onset
before the age of 35. Adolescent girls and young adult women are
most commonly affected. In Western countries, women are 2–3 times
more likely to develop IBS than men, whereas in the Indian
subcontinent men account for 70–80% of IBS patients [5].
Differences in prevalence are partly explained by methodological
factors, including the use of different versions of the Rome
criteria, as well as cultural and healthcare-seeking behaviors.
Geographic variation is evident, with higher prevalence in parts of
Asia and some European countries, while lower rates have been
reported in other regions. The reasons for these differences likely
include hormonal factors, variations in pain perception and
processing, and differences in healthcare utilization patterns.
The syndrome is most commonly diagnosed in individuals under 50
years of age, while prevalence is lower in older populations. This
may reflect spontaneous remission in some patients, as well as
changes in symptom perception or healthcare-seeking behavior. The
highest prevalence has been observed among educated individuals,
higher-income groups, students, and younger populations [6].
Nevertheless, IBS is a universally present disorder regardless of
ethnic or cultural background, confirming its multifactorial nature.
Familial aggregation of IBS suggests a genetic component, but also
the influence of shared environmental factors. Studies show that IBS
is approximately twice as common in first-degree relatives compared
with the general population. However, the exact contribution of
genetic factors versus learned behavioral patterns and shared
stressors remains under investigation.
A significant epidemiological entity is post-infectious IBS.
Following acute gastroenteritis, the risk of developing chronic
IBS-like symptoms is markedly increased. This risk is particularly
pronounced after severe infections, prolonged symptom duration, and
the presence of psychological stressors during recovery. Triggered
by bacterial or viral infections, it involves persistent low-grade
inflammation, altered intestinal permeability, and microbiota
changes, clearly demonstrating the link between inflammation, immune
activation, and long-term alterations in neuromuscular gut function
[7].
Psychosocial factors also have epidemiological importance. Chronic
stress, childhood trauma, and adverse life events are associated
with a higher risk of IBS development and a more severe disease
course. These factors do not act in isolation but rather through
modulation of the brain–gut axis and visceral signal perception [8].
The natural course of IBS is variable. In population studies,
prevalence remains relatively stable over time, but 10–40% of
patients experience symptom remission during long-term follow-up.
IBS rarely progresses to organic disease and is not associated with
increased mortality, but it is linked to significantly reduced
quality of life and increased healthcare resource utilization [9].
Etiopathogenesis
The etiopathogenesis of irritable bowel syndrome (IBS) is not fully
understood, but it is now considered the result of a complex
interaction between genetic, neurobiological, immunological,
microbiological, and psychosocial factors. The contemporary concept
is based on the biopsychosocial model, according to which genetic
predisposition, combined with environmental factors such as acute
gastrointestinal infections, chronic stress, and adverse early-life
events, leads to dysregulation of the brain–gut axis. IBS is
therefore viewed as a disorder of regulation rather than structure,
with pathophysiological mechanisms varying in intensity and
combination among different patient subtypes [10].
One of the central mechanisms is visceral hypersensitivity.
In approximately 60% of patients, a reduced threshold for perception
of mechanical and chemical stimuli in the gastrointestinal tract has
been demonstrated. Peripheral sensitization of enteric neurons, as
well as enhanced central pain processing, leads to increased
perception of otherwise physiological stimuli. Neuroimaging studies
of the central nervous system show increased activation of the
anterior cingulate cortex and amygdala—regions involved in emotional
pain processing—along with reduced activation of the prefrontal
cortex, which is involved in cognitive modulation. These changes
indicate central amplification of visceral signals and explain why
pain in IBS is not proportional to objective findings [11].
Low-grade immune activation represents another important
mechanism. This is particularly evident in post-infectious IBS,
where increased infiltration of T lymphocytes and mast cells in the
intestinal mucosa has been documented. Mast cells, through the
release of histamine, proteases, and cytokines, contribute to
peripheral neuronal sensitization and disruption of epithelial
barrier function. Increased luminal protease activity further
enhances activation of the enteric nervous system. This
microinflammation, although mild, may have long-term effects on
neuromuscular gut function [12].
Disturbances in the serotonergic system also play a
significant role. Serotonin (5-HT), synthesized in enteroendocrine
cells, is a key regulator of intestinal motility, secretion, and
sensory function. In IBS patients, altered serotonin metabolism has
been described, contributing to changes in gastrointestinal transit.
Motor disturbances include increased activity in fasting and
postprandial states, as well as altered responses to stress.
Accelerated transit is present in approximately half of patients
with diarrhea-predominant IBS (IBS-D), while delayed transit is
characteristic of a subset of patients with constipation-predominant
IBS (IBS-C). In about one-quarter of IBS-D patients, increased
colonic bile acid exposure has been observed, contributing to
enhanced secretion and motility, while reduced bile acid
availability is associated with a constipation phenotype [13].
The gut microbiota represents another key element in
pathogenesis. Meta-analyses have demonstrated altered gut microbiota
composition in IBS patients compared with healthy controls, with
reduced diversity and changes in the abundance of specific bacterial
taxa. Dysbiosis may affect serotonin synthesis, immune modulation,
short-chain fatty acid production, and epithelial barrier integrity.
Studies showing symptom improvement after administration of certain
antibiotics or probiotics further support the role of the microbiota
in the syndrome. Increased intestinal permeability, documented in a
subset of patients, represents an important link between stress,
microbiota, and immune activation, allowing increased antigen
translocation and immune sensitization [14].
Psychological factors and stress have a significant
modulatory effect. Chronic stress, childhood trauma, and maladaptive
coping mechanisms—such as catastrophic thinking and persistent fear
and hypervigilance—are associated with increased symptom perception.
Activation of the hypothalamic–pituitary–adrenal (HPA) axis leads to
the release of corticotropin-releasing factor (CRF), which
influences autonomic regulation, motility, secretion, and immune
response. Experimental models show that acute stress increases
visceral sensitivity and intestinal permeability. Dominance of the
sympathetic over the parasympathetic nervous system is more
frequently observed in patients with more severe symptoms, although
a causal relationship remains insufficiently defined [15].
Genetic predisposition represents an additional risk
factor. Twin studies show higher concordance of IBS symptoms in
monozygotic compared to dizygotic twins. Genome-wide analyses have
identified gene variants associated with ion channel function,
autonomic regulation, smooth muscle contractility, and mechanical
sensitivity. In a smaller subset of patients, mutations in the
sucrase–isomaltase gene have been described, leading to carbohydrate
malabsorption and a clinical presentation resembling IBS.
Overall, IBS is a heterogeneous syndrome in which neural,
immunological, microbiological, and psychoneuroendocrine mechanisms
are interrelated. Different combinations and varying dominance of
individual pathophysiological pathways likely explain the clinical
heterogeneity of the syndrome and the variability in therapeutic
response. This understanding of etiopathogenesis opens the
possibility for a personalized therapeutic approach based on
identifying the dominant mechanism in each individual patient.
Diagnostic approach
The modern diagnostic approach to irritable bowel syndrome (IBS) is
based on the concept of establishing a positive clinical diagnosis
rather than relying solely on exclusion. This approach aims to
reduce unnecessary diagnostic procedures, shorten the time to
treatment initiation, and decrease patient anxiety.
Management of IBS requires a thorough medical history, clinical
evaluation, review of previous diagnostic tests, and careful
follow-up. Approximately half of IBS patients present with
extraintestinal symptoms, including headache (23–45%), back pain
(27–81%), fatigue (36–63%), myalgia (29–36%), and urinary symptoms
(21–61%). Extraintestinal symptoms are more common in women,
particularly during menstruation. Depending on symptom severity and
disease burden, patients are divided into three groups according to
an IBS symptom severity scale. Patients with mild symptoms are
usually managed in primary care, as their quality of life is not
significantly impaired. The moderate group is managed at the
secondary care level, while patients with severe symptoms are
typically treated in tertiary care centers [16].
Diagnosis is based on the Rome IV criteria, which require
persistent symptoms over the last 3 months, with symptom onset at
least 6 months prior to diagnosis. The sensitivity and specificity
of these diagnostic criteria are 62.7% and 97.1%, respectively.
The defining feature is recurrent abdominal pain, occurring on
average at least one day per week over the last 3 months, associated
with at least two of the following criteria: (1) related to
defecation, (2) associated with a change in stool frequency, and (3)
associated with a change in stool form/consistency (Figure 1) [9].
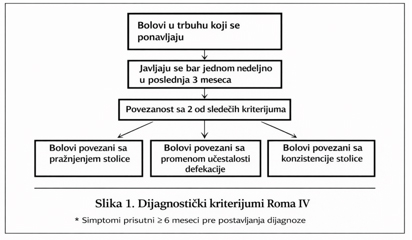
Other symptoms in addition to abdominal pain include: (1)
abnormal stool frequency (<3 bowel movements per week or >3 bowel
movements per day), (2) straining during defecation, (3) urgency or
a sensation of incomplete evacuation, and mucus discharge [17].
According to the predominant symptoms accompanying chronic abdominal
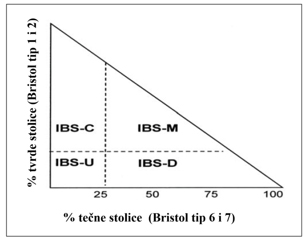
pain over the last 3 months, IBS is classified into four subtypes:
- IBS with predominant constipation (IBS-C) – more than
25% of bowel movements with Bristol Stool Scale types 1 or 2,
and less than 25% with types 6 or 7.
- IBS with predominant diarrhea (IBS-D) – more than 25%
of bowel movements with Bristol Stool Scale types 6 or 7, and
less than 25% with types 1 or 2.
- Mixed IBS (IBS-M) – more than 25% of bowel movements
with Bristol Stool Scale types 1 or 2, and more than 25% with
types 6 or 7.
- Unclassified IBS (IBS-U) – patients fulfilling
diagnostic criteria for IBS that cannot be classified into any
of the above subtypes (Figure 2). [9].
A key component of the initial evaluation is the identification
of alarm symptoms that may indicate organic disease. These include
unexplained weight loss, gastrointestinal bleeding, anemia,
nocturnal symptoms, a family history of colorectal cancer or
inflammatory bowel disease, and onset of symptoms after the age of
50. The presence of these features requires an extended diagnostic
workup, including endoscopic and radiological investigations.
In patients without alarm symptoms, a limited initial laboratory
evaluation is recommended. This typically includes a complete blood
count, C-reactive protein (CRP) or erythrocyte sedimentation rate (ESR),
as well as serological testing for celiac disease, particularly in
patients with diarrhea-predominant symptoms. Fecal calprotectin is a
useful non-invasive biomarker for differentiating IBS from
inflammatory bowel disease, as normal values with high probability
exclude active intestinal inflammation [1].
Endoscopic evaluation is not routinely indicated in younger patients
without alarm features. However, colonoscopy is recommended in
individuals over 50 years of age according to colorectal cancer
screening guidelines, as well as in patients with persistent or
atypical symptoms. In patients with chronic diarrhea, colonic
biopsies should be considered to exclude microscopic colitis, even
in the absence of macroscopic abnormalities.
The differential diagnosis of IBS depends on the dominant
clinical phenotype. In IBS-D, inflammatory bowel disease, celiac
disease, infections, and bile acid malabsorption should be excluded.
In IBS-C, differential diagnoses include primary motility disorders,
hypothyroidism, and drug-induced constipation. Small intestinal
bacterial overgrowth (SIBO) remains a controversial diagnosis but
may be considered in patients with prominent bloating and
refractoriness to standard therapy.
An important aspect of the diagnostic process is the assessment of
psychosocial factors. Anxiety, depression, and chronic stress do not
represent merely comorbidities, but can also modulate symptom
intensity and therapeutic response. Structured evaluation of
psychological burden may help individualize the therapeutic
approach.
Overdiagnosis may have negative consequences, including increased
healthcare costs and heightened health-related anxiety. Therefore, a
rational, stepwise approach is of particular importance. After a
careful medical history, physical examination, and basic laboratory
testing, a reliable diagnosis of IBS can be established in patients
without alarm features, allowing treatment initiation without
additional invasive procedures.
The modern diagnostic concept of IBS thus reflects an understanding
of the syndrome as a disorder of regulation, in which the clinical
symptom pattern is of central importance. This approach enables
faster identification of disease subtypes, reduces unnecessary
interventions, and forms the basis for a personalized therapeutic
plan.
Treatment of Irritable Bowel Syndrome
Therapeutic management of IBS is based on a combination of patient
education, dietary measures, pharmacotherapy, and, when necessary,
psychological interventions. Due to the heterogeneity of
pathophysiological mechanisms, treatment must be individualized and
directed toward the dominant subtype (IBS-C, IBS-D, IBS-M, IBS-U),
as well as the severity of pain, bloating, and psychosocial factors.
General principles of therapy
Patient education is the cornerstone of treatment. Explaining the
benign nature of the condition and the role of the brain–gut axis
reduces anxiety and improves adherence. It is important to emphasize
that IBS has a chronic, fluctuating course and that the goal of
therapy is symptom control and improvement of quality of life,
rather than complete cure.
Dietary measures: The assumption that IBS symptoms are
strictly diet-related is often overestimated, and elimination diets
and fiber supplementation rarely provide long-term symptom
improvement. A low-FODMAP diet, which restricts fermentable
oligosaccharides, disaccharides, monosaccharides, and polyols, may
improve symptoms in about one in five patients by reducing bloating
and abdominal pain [18].
Individual assessment of intolerance to lactose, fructose, and other
fermentable carbohydrates may be useful. However, overly restrictive
diets should be avoided due to the risk of nutritional deficiencies
and negative effects on the gut microbiota.
Lifestyle modifications (regular physical activity,
adequate sleep, and stress management) may contribute to symptom
relief. In patients with moderate to severe symptoms, a combination
of pharmacological and non-pharmacological approaches is often
required [9].
Pharmacological treatment of IBS-C (constipation-predominant
IBS)
The goals of therapy are to increase stool frequency and improve
stool consistency, while reducing pain and bloating. Fiber (psyllium,
methylcellulose, calcium polycarbophil) is first-line therapy.
Meta-analyses show moderate efficacy. Wheat bran has not
demonstrated significant benefit.
Osmotic laxatives (PEG, magnesium-containing mineral waters) improve
stool consistency and reduce straining, but have limited effect on
pain and bloating. Lactulose and sorbitol are not recommended due to
bloating. Stimulant laxatives (senna, bisacodyl) are used
short-term.
In patients with persistent symptoms, secretagogues and pro-secretory
agents are used:
Lubiprostone (ClC-2 channel activator) improves stool
consistency and reduces pain; common adverse effects include nausea
and diarrhea.
Plecanatide (guanylate cyclase-C agonist) increases
intestinal secretion and reduces visceral hypersensitivity, with
beneficial effects on abdominal pain.
Tenapanor (Na⁺/H⁺ exchanger inhibitor) increases stool water
content and reduces abdominal pain.
Tegaserod (5-HT4 agonist) may be considered in women under 65
years without cardiovascular risk.
Prucalopride, a selective 5-HT4 receptor agonist, stimulates
colonic peristalsis and is particularly useful in chronic
constipation with poor response to previous treatments.
For prominent abdominal pain and visceral hypersensitivity,
especially in the presence of anxiety or depression, SSRIs
(selective serotonin reuptake inhibitors) or SNRIs (serotonin–norepinephrine
reuptake inhibitors) may be used, while TCAs (tricyclic
antidepressants) can be beneficial in patients with pain and slow
intestinal transit.
Irritable Bowel Syndrome (IBS)
Irritable bowel syndrome (IBS) is a chronic gastrointestinal
disorder characterized by recurrent abdominal pain associated with
changes in stool frequency and/or stool consistency. Symptoms vary
in intensity and frequency. IBS is classified into subtypes: IBS-D (diarrhea-predominant),
IBS-C (constipation-predominant), mixed IBS (IBS-M), and
unclassified IBS (IBS-U).
The etiopathogenesis is multifactorial, including visceral
hypersensitivity, alterations in central nervous system processing
of signals, motility disturbances, immune activation, microbiota
dysbiosis, and increased intestinal permeability. Psychological
factors, stress, and childhood trauma further modify symptom
expression. The prevalence of IBS is estimated at 10–15% of the
general population, with higher frequency in women and individuals
younger than 50 years.
Genetic and familial factors contribute to susceptibility, while
acute gastroenteritis and post-infectious forms represent important
etiological triggers. Epidemiological data suggest a relatively
stable prevalence over time, with spontaneous remission occurring in
a subset of patients. IBS does not increase mortality but is
associated with reduced quality of life and increased healthcare
utilization.
Biopsychosocial Model and Clinical Significance
Modern understanding of IBS shifts the focus from purely
gastrointestinal symptoms toward a broader concept of brain–gut axis
dysregulation. Interactions between the enteric nervous system,
autonomic regulation, microbiota, immune responses, and
psychological factors shape the individual disease phenotype. This
explains why uniform therapeutic approaches often yield limited
results and highlights the importance of personalized treatment
strategies.
Although IBS does not lead to structural intestinal damage or
increased mortality, it represents a significant public health
burden due to frequent medical consultations, diagnostic procedures,
reduced productivity, and impaired quality of life. Psychological
distress, stigma, and the chronic nature of symptoms further
contribute to disease burden.
IBS should therefore not be viewed as a single disease entity, but
rather as a heterogeneous clinical syndrome with varying dominant
mechanisms, allowing for more precise diagnostic and therapeutic
approaches.
Epidemiology
IBS is one of the most common gastrointestinal disorders in the
general population. Population-based studies estimate a prevalence
of 10–15% and an annual incidence of 1–2%.
Prevalence varies significantly across countries, ranging from 1.1%
in France to 35.5% in Mexico, while a pooled prevalence of 7.1% has
been reported in the USA, Europe, Australia, and New Zealand.
Approximately 50% of patients report symptom onset before the age of
35.
IBS is more common in young adult women; in Western countries, women
are 2–3 times more likely to be affected than men, whereas in some
regions (e.g., the Indian subcontinent), men may predominate among
diagnosed cases.
The syndrome is most frequently diagnosed in individuals under 50
years of age. Familial aggregation suggests a genetic contribution,
although shared environmental factors also play a significant role.
Post-infectious IBS is an important subtype, developing after acute
gastroenteritis and involving low-grade inflammation, altered
permeability, and microbiota changes.
Psychosocial factors such as chronic stress and early-life trauma
significantly influence both risk and symptom severity. The natural
course is variable, with symptom remission in 10–40% of patients,
while mortality is not increased.
Etiopathogenesis
The etiopathogenesis of IBS is not fully understood, but it is
considered the result of complex interactions among genetic,
neurobiological, immunological, microbiological, and psychosocial
factors. The biopsychosocial model explains IBS as a disorder of
regulation rather than structure.
Visceral hypersensitivity is a key mechanism, present in
approximately 60% of patients, involving both peripheral
sensitization and altered central pain processing. Neuroimaging
studies show increased activation of the anterior cingulate cortex
and amygdala, with reduced prefrontal modulation.
Low-grade immune activation is particularly relevant in
post-infectious IBS, with increased mast cells and T lymphocytes
contributing to neural sensitization. Serotonergic dysregulation
affects motility and secretion, while bile acid malabsorption
contributes to diarrhea in IBS-D.
Alterations in gut microbiota (dysbiosis) influence immune
activation, serotonin metabolism, and epithelial barrier integrity.
Increased intestinal permeability may allow antigen translocation
and immune activation.
Psychological factors, including chronic stress and early-life
trauma, modulate symptoms via the hypothalamic–pituitary–adrenal (HPA)
axis, leading to altered motility, secretion, and visceral
sensitivity.
Genetic studies suggest heritability, with involvement of genes
related to ion channels, smooth muscle function, and autonomic
regulation.
Diagnosis
The modern diagnostic approach is based on a positive diagnosis
using Rome IV criteria, rather than exclusion alone. IBS is defined
by recurrent abdominal pain occurring at least 1 day per week over
the past 3 months, associated with at least two of the following:
relation to defecation, change in stool frequency, or change in
stool form.
Alarm features (weight loss, bleeding, anemia, nocturnal symptoms,
family history of colorectal cancer/IBD, or onset after age 50)
require further investigation.
Basic evaluation includes blood tests (CBC, CRP/ESR), celiac
serology, and fecal calprotectin. Endoscopy is not routinely
required in young patients without alarm features.
IBS is classified into IBS-C, IBS-D, IBS-M, and IBS-U based on stool
patterns (Bristol Stool Scale).
Psychosocial assessment is an important part of diagnosis, as
anxiety, depression, and stress significantly influence symptom
severity.
Treatment
Treatment is individualized and based on symptom predominance.
General principles
Education is essential, emphasizing the benign but chronic nature of
IBS and the role of the brain–gut axis. Dietary and lifestyle
modifications include physical activity, sleep regulation, and
stress management.
A low-FODMAP diet may improve symptoms in approximately 20% of
patients. Excessively restrictive diets should be avoided.
IBS-C (Constipation-predominant)
First-line therapy includes dietary fiber (psyllium,
methylcellulose). Osmotic laxatives (PEG) improve stool consistency.
Secretagogues and prokinetics include lubiprostone, plecanatide,
tenapanor, tegaserod, and prucalopride.
Antidepressants (SSRIs, SNRIs, TCAs) may be used for pain and
visceral hypersensitivity.
IBS-D (Diarrhea-predominant)
Loperamide is first-line therapy. Eluxadoline reduces
diarrhea and pain but is contraindicated in patients without a
gallbladder.
Rifaximin improves bloating and global symptoms. Bile acid
sequestrants are used in suspected bile acid malabsorption.
5-HT3 antagonists (alosetron, ondansetron, ramosetron) are
used in refractory cases.
Antispasmodics and neuromodulators are also effective for pain.
IBS-M and IBS-U
Treatment is symptom-driven and flexible. Neuromodulators and
antispasmodics play a central role. Probiotics (especially
Bifidobacterium strains) may improve symptoms in a subset of
patients.
Neuromodulators and Psychological Therapy
Neuromodulators (TCAs, SSRIs, SNRIs) act on the brain–gut axis and
reduce visceral hypersensitivity.
Psychological therapies include cognitive-behavioral therapy (CBT),
gut-directed hypnotherapy, relaxation techniques, and stress
management programs. CBT is the most extensively studied and has
demonstrated significant efficacy in reducing symptom severity and
improving quality of life..
Gut-directed hypnotherapy acts through central mechanisms of
modulation of visceral perception. Studies demonstrate clinically
significant improvement in symptoms after 6–12 weeks of therapy.
Relaxation techniques may improve quality of life, although their
effect on core IBS symptoms is less consistent.
Due to the chronic course of the disease, many patients turn to
complementary medicine. Acupuncture, according to available
analyses, is likely not superior to placebo. Evidence for herbal
treatments is limited, although some studies suggest potential
benefit from individualized approaches.
IBS treatment must be personalized. The foundation consists of
patient education, rational dietary intervention, and targeted
pharmacotherapy. In cases of pronounced central sensitization and
psychological comorbidities, the combination of neuromodulators and
psychological interventions often yields the best results. This
individualized and combined approach reflects the modern
understanding of IBS as a heterogeneous disorder of brain–gut axis
regulation and enables optimal symptom control with minimal
therapeutic burden for the patient [1]. A schematic overview of the
therapeutic approach for IBS-C and IBS-D is shown in Figure 3.

CONCLUSION
Irritable bowel syndrome (IBS) is a common and chronic functional
gastrointestinal disorder that significantly impairs patients’
quality of life. In patients who fulfill the Rome IV diagnostic
criteria and do not present alarm features, the diagnosis can be
established based on a carefully obtained medical history, analysis
of typical clinical symptoms, and physical examination, without the
need for extensive diagnostic testing. In contrast, the presence of
alarm symptoms, onset after the age of 50, a positive family history
of colorectal cancer, or suspicion of celiac disease or chronic
inflammatory bowel disease requires additional laboratory, imaging,
and endoscopic evaluation.
Management of IBS begins with a clear explanation of the nature of
the disorder and reassurance that it is not a malignant or
life-threatening condition. Treatment is long-term and
individualized, targeting predominant symptoms and their severity.
The foundation of therapy includes patient education, dietary
measures, and targeted pharmacotherapy, while neuromodulators and
psychological interventions play an important role in patients with
pronounced visceral hypersensitivity and psychological comorbidities.
Successful management requires continuous and constructive
collaboration between the patient and the physician, with
involvement of other specialists when necessary. A multidisciplinary
and personalized approach reflects the modern understanding of IBS
as a disorder of brain–gut axis regulation and enables optimal
symptom control while preserving quality of life.
Literature:
1. Huang KY, Wang FY, Lv M, Ma XX, Tang XD, Lv L. Irritable bowel
syndrome: Epidemiology, overlap disorders, pathophysiology and
treatment. World J Gastroenterol. 2023 Jul 14;29(26):4120-4135
2. Videlock EJ, Chang L. Irritable bowel syndrome, In: Wang TC,
Camilleri M, eds. Yamada’s Textbook of Gastroenterology: Seventh
edition. New York: Wiley; 2022. p. 1374–407.)
3. Chey WD, Kurlander J, Eswaran S: Iritable bowel syndrome: a
clinical review. JAMA 2015;313:945–58.
4. Ford AC, et al. Evidence-based clinical practice guidelines for
irritable bowel syndrome. Nat Rev Gastroenterol Hepatol.
2020;17:458–476.
5. Canavan C, West J, Card T. The epidemiology of irritable bowel
syndrome. Clin Epidemiol. 2014. 6:71-80.
6. Husain N, Chaudhry IB, Jafri F, Niaz SK, Tomenson B, Creed F. A
population-based study of irritable bowel syndrome in a non-Western
population. Neurogastroenterol Motil. 2008;20:1022–1029. doi:
10.1111/j.1365-2982.2008.01143.x. ).
7. Thabane M, Marshall JK. Post-infectious irritable bowel syndrome.
World J Gastroenterol. 2009 Aug 7;15(29):3591-6.)
8. Ballou S, Bedell A, Keefer L. Psychosocial impact of irritable
bowel syndrome: A brief review. World J Gastrointest Pathophysiol.
2015 Nov 15;6(4):120-3.
9. Štabuc B. Sindrom razdražljivega črevesa - najpogosteša
funkcionalna črevesna bolezen Slovenian Journal of Gastroenterology
/ Gastroenterolog 2024; 3: 52–6
10. Tang HY, Jiang AJ, Wang XY, et al. Uncovering the
pathophysiology of irritable bowel syndrome by exploring the
gut-brain axis: a narrative review. Ann Transl Med. 2021 Jul.
9(14):1187).
11. Van den Houte K, Bercik P, Simren M, Tack J, Vanner S.
Mechanisms underlying food-triggered symptoms in disorders of
gut-brain interactions. Am J Gastroenterol. 2022 Jun 1.
117(6):937-46.
12. Gao J, Xu K, Liu H, et al. Impact of the gut microbiota on
intestinal immunity mediated by tryptophan metabolism. Front Cell
Infect Microbiol. 2018. 8:13. [
13. 13 Camilleri M. Physiological underpinnings of irritable bowel
syndrome: neurohormonal mechanisms. J Physiol. 2014 Jul 15.
592(14):2967-80
14. Kim GH, Lee K, Shim JO. Gut bacterial dysbiosis in irritable
bowel syndrome: a case-control study and a cross-cohort analysis
using publicly available data sets. Microbiol Spectr. 2023 Feb 14.
11(1):e0212522.
15. Ng QX, Yaow CYL, Moo JR, Koo SWK, Loo EXL, Siah KTH. A
systematic review of the association between environmental risk
factors and the development of irritable bowel syndrome. J
Gastroenterol Hepatol. 2024 May 3.
16. Francis CY, Morris J, Whorwell PJ. The irritable bowel scoring
system: a simple method of monitoring IBS and its progress. Aliment
Pharmacol Therapeut 1997;11:395–402.
17. Palsson OS, Whitehead WE, Miranda AL, Chang L, Chey W, Crowel
MD. Development and validation of the Rome IV diagnostic
questionnaire for adults. Bowel disorders. Gastroenterology
2016;150:1481–91.)
18. Bijkerk CJ, de Wit NJ, Muris JWM, Whorwell PJ, Knottnerus JA,
Hoes AW. Soluble or insoluble fibre in irritable bowel syndrome in
primary care? Randomised placebo controlled trial. BMJ
2009;339:b3154.
|
|
|
|



